Category Archives: chemistry in everyday life
Structure-based drug design updates for SARS-CoV-2
I will be posting here information that is published regarding available crystal structures from SARS-CoV-2 and any other information relevant to structure-based drug design against the virus. Key proteins and their roles in viral infection can be found here. I hope you find this informative and useful for your research.
25 March 2020: 68 crystal structures of SARS-CoV-2 protease, with various bound fragments identified using PanDDA analysis are listed here by EBI.
25 March 2020: The Diamond Light Source (UK) has been able to solve a new structure of the SARS-CoV-2 main protease (MPro) at high resolution (1.25 Å, PDB ID: 6YB7), and subsequently complete a large XChem crystallographic fragment screen against it (detailed here). Data have been deposited with the PDB, and are made available immediately to the world on this page; additional work is ongoing, and updates will be continually posted in their website. On Tuesday March 17th they publicly released results from the full 1500-crystal experiment that yielded 58 non-covalent and covalent active-site fragments. Following data reprocessing and further analysis, an additional 13 structures were released on March 24th taking the final total to 66 active site fragments, 44 of which were covalently bound (full timeline here, download page here). This was an exceptionally large screen, and yielded an exceptionally rich readout, with vast opportunities for fragment growing and merging.
25 March 2020: Blog post, with code by Patrick Walters in Practical Cheminformatics “Building on the Fragments From the Diamond/XChem SARS-CoV-2 Main Protease (MPro) Fragment Screen (Part I)” . Share schemoinformatics techniques and the code used when working with the results of fragment screens.
25 March 2020: Visualize SILCS FragMaps for the SARS-CoV-2 main protease (PDB ID 6LU7) through the SILCS demo web viewer, and check out the 6LU7 crystallographic ligand and the XChem fragment screen results from the Diamond Light Source.
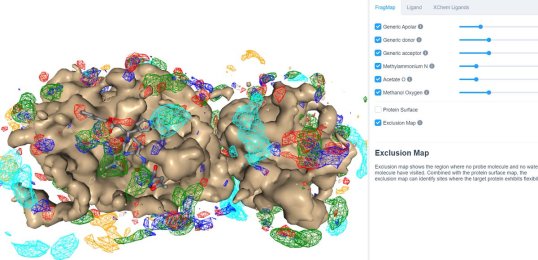
24 March 2020: Results of Diamond Light Source fragment screen:There were 68 hits of high interest – data and extensive details are here, and some interactive views here:
- 22 non-covalent hits in the active site
- 44 covalent hits in the active site
- 2 hits in the dimer interface, one in a calculated hotspot
Based on these data, the team invites chemists around the world to design new compounds or present existing compounds that could bind to the protease and submit them here.Structures submitted are prioritized by factors such as ease of synthesis and potential toxicity and the compounds selected will be synthesized and evaluated for binding to the SARS-CoV-2 protease.
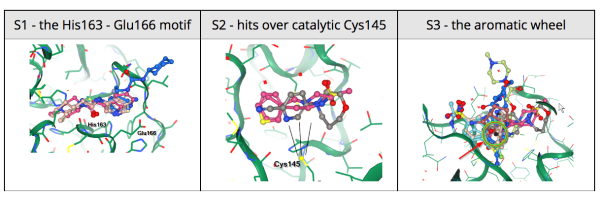 Results from the XChem fragment screen of the Diamond Light Source can be found here.
Results from the XChem fragment screen of the Diamond Light Source can be found here.
20 March 2020: The crystal structure of SARS-CoV-2 main protease is published complexed with an α-ketoamide inhibitor (PDB ID: 6Y7M). The main viral protease (Mpro, also called 3CLpro) is one of the best characterized drug targets among coronaviruses. 3CLpro protease is required for the virus but lacks human homologous proteins and thus inhibitors of this protease are less likely to bind to a human protease. Research teams in Germany have been able to crystallize the protease and have used this structure to optimize an existing α-ketoamide inhibitor developed to combat other diseases.
 SARS-CoV-2 coronavirus protease dimer bound to an α-ketoamide inhibitor (yellow). The image is reproduced from C&EN news.
SARS-CoV-2 coronavirus protease dimer bound to an α-ketoamide inhibitor (yellow). The image is reproduced from C&EN news.
20 March 2020: SwissProt has modeled the full SARS-CoV-2 proteome based on the NCBI reference sequence NC_045512 which is identical to GenBank entry MN908947, and annotations from UniProt. All data is deposited here.
20 March 2020: Check out refined coordinates of existing experimental SARS-CoV-2 structures using ISOLDE by Tristan Croll (Cambridge University). Modeled coordinates are deposited here.
20 March 2020: Prediction of 10 models for SARS-CoV-2 proteins from the Feig lab.
17 March 2020: The first vaccine clinical trial against SARS-CoV-2 starts in Seattle by Moderna in collaboration with NIAID. The experimental vaccine contains synthetic m-RNA clones that encode glycoprotein S. Once human cells recognize the genetic material of the virus, it is hoped that they will produce antibodies that inactivate the viral glycoprotein S. This strategy is different from the Influenza vaccine, where the viral surface protein itself, hemagglutinin, is introduced into the body and the human immune system produces antibodies against that protein.
5 March 2020: Check out computational predictions of protein structures associated with COVID-19 by DeepMind using AlphaFold, their recently published deep learning system, focuses on predicting protein structure accurately when no structures of similar proteins are available, called “free modelling”.
3 March 2020: Researchers at the Structural Genomic Infectious Diseases Center resolve the crystal structure of SARS-CoV-2 Nsp15 / NendoU endoribonuclease in high resolution. (PDB IDs: 6W01, 6VWW).
20 February 2020: A third research team also published the structure of the SARS-CoV-2 glycoprotein S of SARS-CoV-2 in two conformations using cryo-electron microscopy. (PDB IDs:6VXX, 6VYB)
19 February 2020: A research team from China presents cryo-EM structures of the SARS-CoV-2 glycoprotein S bound to the human ACE2 receptor. The structure shows the full-length human ACE2, in the presence of a neutral amino acid transporter B0AT1, with or without the receptor binding domain (RBD) of the surface spike glycoprotein (S protein) of SARS-CoV-2, both at an overall resolution of 2.9 Å, with a local resolution of 3.5 Å at the ACE2-RBD interface (PDB IDs: 6M18, 6M1D, 6M17).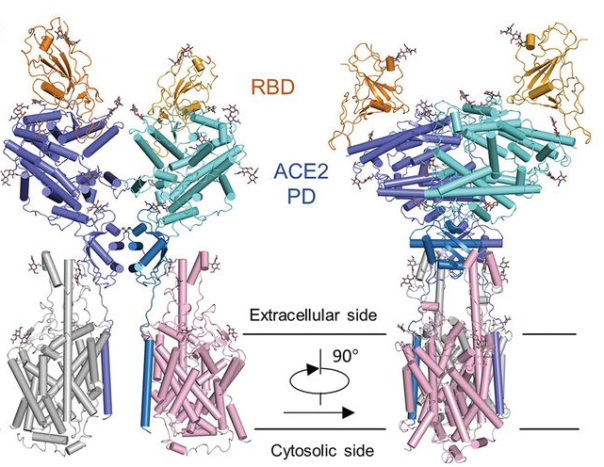 Overall structure of the RBD-ACE2-B0AT1 complex. The complex is colored by subunit, with the ACE2 protease (PD) region depicted in cyan and the Collectrin-like region (CLD) of ACE2 in blue. The glycosylated parts of the human ACE2 receptor are illustrated in strick representation. The RBD subunit of the structure of glycoprotein S that binds to ACE2 is depicted in yellow. The figure has been adapted from the original publication.
Overall structure of the RBD-ACE2-B0AT1 complex. The complex is colored by subunit, with the ACE2 protease (PD) region depicted in cyan and the Collectrin-like region (CLD) of ACE2 in blue. The glycosylated parts of the human ACE2 receptor are illustrated in strick representation. The RBD subunit of the structure of glycoprotein S that binds to ACE2 is depicted in yellow. The figure has been adapted from the original publication.
15 February 2020: The first cryo-EM structure of the viral spike protein (glycoprotein S) is published at a resolution of 3.5 Å (PDB code: 6VSB). Based on the SARS-CoV-2 genome sequence shared by Chinese researchers, they managed to prepare a purified sample of the spike protein and to determine its structure using single particle cryo-EM in less than two weeks. Using surface plasmon resonance they also measured that SARS-CoV-2 glycoprotein S binds 10-20 times more to the human cell ACE2 receptor than the 2002 SARS-CoV virus glycoprotein S. 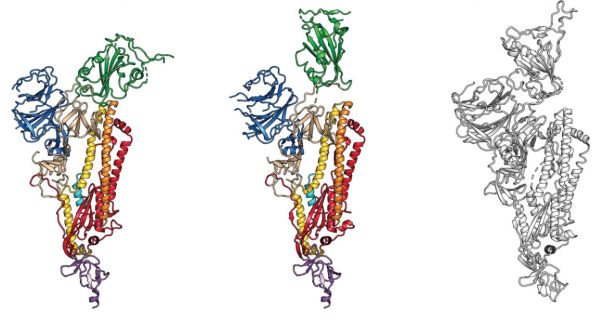 The structure of the glycoprotein S of SARS-CoV-2, as identified by cryo-electron microscopy. Green binds to the binding site that binds to human cells. In the middle image, the green binding domain of glycoprotein S is positioned for binding to the human enzyme ACE2. On the right is the SARS-CoV glycoprotein S for comparison. The figure has been adapted from the original publication.
The structure of the glycoprotein S of SARS-CoV-2, as identified by cryo-electron microscopy. Green binds to the binding site that binds to human cells. In the middle image, the green binding domain of glycoprotein S is positioned for binding to the human enzyme ACE2. On the right is the SARS-CoV glycoprotein S for comparison. The figure has been adapted from the original publication.
5 February 2020: The crystal structure of the SARS-CoV-2 main protease is released in the PDB (PDB ID: 6LU7).
3 February 2020: The complete viral genome of the new coronavirus was analyzed by scientists in China.
30 January 2020: The World Health Organization has named the new virus SARS-CoV-2 and the disease caused by the new virus, COVID-19, and the outbreak was declared a Public Health Emergency of International Concern.
5 January 2020: The source of this infection was quickly identified as a new coronavirus that resembles those that caused SARS-CoV outbreaks in 2002-2004 and MERS-CoV in 2012, but is not the same virus.
31 December 2019: A pneumonia of unknown cause detected in Wuhan, China was first reported to the WHO Country Office in China on 31 December 2019.
How are drugs designed?
How are drugs designed? Come on Friday 2nd May at 18:00 at the Athens Science Festival, Technopolis Gkazi to listen to my talk! See the detailed program of the festival here.
 This talk will address how drugs are designed to combat a disease – from the discovery of the cause of the disease (e.g. a mutant protein), specialized techniques for the design of small chemical molecules (that are drugs), to clinical trials. We will also discuss advances and progresses in individualized treatment (also known as personalized medicine), i.e. how DNA testing could help each patient receive medication specifically tailored for them.
This talk will address how drugs are designed to combat a disease – from the discovery of the cause of the disease (e.g. a mutant protein), specialized techniques for the design of small chemical molecules (that are drugs), to clinical trials. We will also discuss advances and progresses in individualized treatment (also known as personalized medicine), i.e. how DNA testing could help each patient receive medication specifically tailored for them.
Πώς σχεδιάζονται τα φάρμακα; Ελάτε την Παρασκευή 2 Μαϊου στις 18:00 στο Athens Science Festival, Τεχνοπολις Γκάζι, να ακούσετε την ομιλία μου.
Η ομιλία έχει θέμα το πώς επιτελείται ο σχεδιασμός φαρμάκων για μία ασθένεια – από την ανακάλυψη του αιτίου που προκαλεί την ασθένεια (π.χ. μια μεταλλαγμένη πρωτεΐνη), εξειδικευμένες τεχνικές για το σχεδιασμό μικρών χημικών μορίων που αποτελούν τα φάρμακα, μεχρι και τις κλινικές δοκιμές. Επίσης θα συζητήσουμε τις προοπτικές και εξελίξεις στην εξατομικευμένη θεραπεία, το πώς δηλαδή με εξέταση DNA μπορεί ο κάθε ασθενής να λαμβάνει ένα φάρμακο που είναι ειδικά κατάλληλο για εκείνον.
Περισσότερες πληροφορίες και μία μικρή περίληψη για το θέμα θα βρείτε σε πρόσφατο άρθρο στην ιστοσελίδα της διοργάνωσης.
Pheromones are in the Air: Friends, Lovers or Foes
Love at first sight? Desire in a bottle? There have been a lot of claims about what pheromones can do, but what is the science? Dr. George Preti and Dr. Charles J. Wysocki of the Monell Chemical Senses Center discuss the chemistry of pheromones and how mood and emotion can be communicated through chemical signals.
Kalydeco: The most important new drug of 2012
Kalydeco, a drug for cystic fibrosis, is the most important new drug of 2012 according to Forbes magazine and was developed by Vertex pharmaceuticals with seed funding from the Cystic Fibrosis Foundation.
Cystic Fibrosis is a genetic disorder that results in scarring (fibrosis) and cyst formation within the pancreas, lungs, liver, and intestines.
Kalydeco, given alone, will only help a few thousand patients the world over. Like other drugs for very rare diseases, its price is very high: $294,000 per patient per year.
Though its chemical structure could be routinely made by a synthetic chemist, it is covered by a patent so it is illegal to make in a lab.
The efforts to cure cystic fibrosis were spearheaded by a discovery from Francis Collins, later famous for heading the Human Genome Project and then the National Institutes of Health, who discovered the gene that, when mutated, causes cystic fibrosis 23 years ago. Kalydeco is the first drug to directly affect the defects caused by these mutations, leading to improvements in patients’ lung function.
The Chemistry of Euro banknotes
Europium(III) oxide, alongside other chemicals, is phosphorescent and is used in the anti-counterfeiting details in Euro banknotes.
 Europium (symbol Eu) is one of the rare earth elements and belongs to the class of “lanthanides”.
Europium (symbol Eu) is one of the rare earth elements and belongs to the class of “lanthanides”.
Most of the trivalent rare earth elements are luminescent. This means that they can be excited by shining a light of a particular wavelength at them. When the ions relax again, they emit light – of a different wavelength. That is luminescence.
Euro notes luminesce in the red, green and blue (excited by 254 nm). The red light is clearly linked to europium and most likely to a Eu3+-β-diketone complex.
ChemMatters: Demystifying Everyday Chemistry for high school students
ChemMatters is a magazine for high school students and high school teachers published by the Education Division of the American Chemical Society.
The magazine, which appears four times a year, contains articles that feature real-world applications of chemistry concepts introduced in the classroom. Latest issues cover themes such as the application of nanotechnology, great discoveries in chemistry, and the science behind weather folklore.
The site provides a Teacher’s guide, videos, material for the classroom and more.
Check out the ChemMatters web site for the latest issue!
View the lastest Episode on ChemMatters: “Episode 10: Graphene: The Next Wonder Material?” on carbon allotropes and the applications of graphene.
The Chemistry and Physics of Tennis
Tennis has grown to be my favorite sport, so I found this great YouTube video of Dr. Ainissa Ramirez from Yale University explaining the science behind tennis:
Basically, modern rackets are high tech composits of carbon fibers embedded in epoxy (glue), which give them high strength and low weight. The most expensive strings are made of cow gut, but most commonly nylon is used, which is a flexible polymer.

- Nylon, a synthetic polymer used for tennis racket strings.
Strings hit the ball and create topspin, which makes the ball fall sooner than a ball without top spin. This effect is called the “Magnus effect“. This is what happens to a ball with topspin: while the ball is rotaitng, a thin layer or air around it is also rotating with it. That means that the velocity of air below it is faster than the velocity of air above it, causing it to fall sooner on the ground. That’s why you have to use top spin with your balls if you want to be too fast for your opponent.
Venus Williams usually hits a 1,200 rpm ball and Rafa Nadal 3,600 rpms. That’s faster than a revolving washing machine:-)
After reading some tennis science, I hope you agree that knowing a little bit about the chemistry and physics of tennis can really improve you as a player…! 🙂
Alcohol: Drink, Chemical, Medicine, Poison
What Is Alcohol?
When you mention “alcohol”, you most certainly mean “ethanol”, a simple chemical which is created when grains, fruits, or vegetables are fermented. Fermentation is a process that takes place when yeast (fungi) convert food sugars into ethanol and carbon dioxide.
Sugar + yeast —> alcohol + carbon dioxide
Or
C6H12O6 + yeast ——> CH3CH2OH + CO2
Drink
 So what happens in your body when you start sipping this delicious martini????
So what happens in your body when you start sipping this delicious martini????
- Alcohol travels to your stomach.
- From the stomach, it is quickly passed into the bloodstream, where it travels in a few minutes to every part of the body, including your brain.
- In the brain alcohol affects your neurons: It alters neuron’s membranes and ion channels, enzymes such as acetylcholine, serotonin, and GABA receptors.
- When alcohol binds to these enzymes, it slows the function of the central nervous system and that’s why you feel more relaxed and stress-free.
- Ethanol also inhibits the production of a hormone (ADH) that regulates urine flow, causing increased urine production and dehydration.
- Alcohol increases dopamine by stopping enzymes that degrade it – that’s why you feel happier!
- Ethanol also causes blood vessels to dilate, resulting in flushing of the skin and a sensation of warmth as blood moves into capillaries.
- If you’ve had a large meal, alcohol will stay longer in your stomach, that’s why you can’t get drunk as easily. But eventually, it will enter the bloodstream and do its tricks:)
Your body sees alcohol as an intruder and tries to get rid of it using an enzyme to break it down, “alcohol dehydrogenase“. On contact, the enzyme snatches a hydrogen atom off the ethanol molecules in your drink, rendering it into non-intoxicating acetaldehyde, which is associated with hangovers. Alcohol dehydrogenase is less efficient in Asian populations. Lungs and liver also remove 10% of the alcohol in your body by urine and breath.
Click on this link for a schematic representation of how alcohol works in your brain.
Chemical
- Fuel: The largest single use of ethanol is as a motor fuel, fuel additive, and as a rocket fuel. Over 20% of cars in Brazil are able to run on 100% ethanol fuel.
- Raw material: Ethanol is an important industrial ingredient and has widespread use as a base chemical for other organic compounds, such as ethyl halides, ethyl esters, diethyl ether, acetic acid.
- Solvent: Ethanol is miscible with water and is a good general purpose solvent. It is found in paints, tinctures, markers, and personal care products such as perfumes and deodorants.
- Preservative: for biological specimens.
Medicine
- Antiseptic: Ethanol is used in antiseptic and some antibacterial soaps and wipes. Ethanol kills organisms by denaturing their proteins and dissolving their lipids and is effective against most bacteria and fungi, and many viruses, but is ineffective against bacterial spores.
- Solvent: As a good solvent, you will find ethanol frequently used in many medicine, such as cough syrups.
- Drug: For therapeutic neurolysis: It is injected proximate to nerve tissues and into spinal subarachnoid spaces to produce degeneration of nerve function (neurolysis) for control of chronic pain.
Poison
A couple of days ago I heard in the news that Amy Winehouse‘s death this summer was the result of alcohol poisoning….
A pathologist told a coroner’s court in north London that alcohol toxicity was the cause of the 27-year-old’s death, with her blood-alcohol levels measured at more than five times the legal limit for driving.
The report said that Amy’s blood alcohol content was 416 mg per decilitre at the time of her death. That’s 0.416% alcohol in the blood and about 9 drinks for a woman 45 kg like Amy. When large amounts of alcohol are consumed in a short period of time, alcohol poisoning can occur. Above a blood alcohol concentration of 0.5%, alcohol depresses nerves that control involuntary actions such as breathing and cardiovascular regulation. Moreover, it can cause hypothermia (low body temperature), hypoglycemia (too little blood sugar) and sever dehydration. These effects can lead to cause seizures, permanent brain damage, and ultimately death. That’s why you should never underestimate the effects of alcohol. And think twice before sipping more than three drinks in a row! Better to be safe than sorry.
How is coffee decaffeinated?
Apparently it’s National Coffee Day today, read below to learn in less than a page how coffee gets decaffeinated – not an easy task!
Coffee contains over 400 chemicals important to the taste and aroma of the final drink: it is therefore challenging to remove only caffeine while leaving the other chemicals at their original concentrations.
 To get rid of the caffeine, unroasted (green) beans are at first steamed. The beans are then rinsed with a “solvent” chemical that extracts the caffeine while leaving the other essential chemicals in the coffee beans. The process is repeated anywhere from 8 to 12 times until caffeine is removed in 97%-99.9%. So what is this magic “solvent” chemical. which is able to selectively remove caffeine while preserving the coffee aroma?
To get rid of the caffeine, unroasted (green) beans are at first steamed. The beans are then rinsed with a “solvent” chemical that extracts the caffeine while leaving the other essential chemicals in the coffee beans. The process is repeated anywhere from 8 to 12 times until caffeine is removed in 97%-99.9%. So what is this magic “solvent” chemical. which is able to selectively remove caffeine while preserving the coffee aroma?
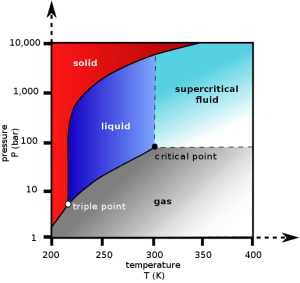
This solvent is nothing else but carbon dioxide (CO2), which is also found in our very own atmosphere. However, as you might have guessed, CO2 is not used for decaffeination as a gas from the atmosphere. For the decaffeination process, the coffee beans are soaked in a special form of CO2 called “supercritical” CO2. OK, so now you naturally have to ask what the heck is this “supercritical” CO2??
Every substance (water, hydrogen, oxygen, etc) has a so-called “critical point”. The critical point is the point above which, the distinction between liquid and gas phases of that substance stops being meaningful. That is you cannot tell any more whether this substance is gas or liquid. This new “phase”, being neither gas or liquid is called “supercritical”. A “supercritical fluid” can effuse through solids like a gas, and dissolve materials like a liquid.
So how does supercritical CO2 look like?
To achieve the supercritical state for most substances requires extreme temperatures and pressures. Carbon dioxide, however, has a fairly accessible critical point at ~ 31.1°C and 73 atm (our atmosphere has 1 atm). And it seems that supercritical CO2 has unique properties such that it can selectively extract caffeine from coffee beans, producing decaf coffee.
Carbon dioxide pressure-temperature phase diagram. The critical point is found at T=31.1°C and 73 atm. Above the critical point, one cannot really tell whether CO2 is liquid or gas. This new phase is called “supercritical”.