Category Archives: materials
ChemMatters: Demystifying Everyday Chemistry for high school students
ChemMatters is a magazine for high school students and high school teachers published by the Education Division of the American Chemical Society.
The magazine, which appears four times a year, contains articles that feature real-world applications of chemistry concepts introduced in the classroom. Latest issues cover themes such as the application of nanotechnology, great discoveries in chemistry, and the science behind weather folklore.
The site provides a Teacher’s guide, videos, material for the classroom and more.
Check out the ChemMatters web site for the latest issue!
View the lastest Episode on ChemMatters: “Episode 10: Graphene: The Next Wonder Material?” on carbon allotropes and the applications of graphene.
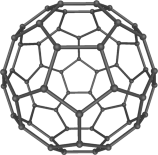
Happy Halloween! (with a fullerene twist)
The 1996 Nobel Prize for Chemistry has been won by Harold W. Kroto, Robert F. Curl and Richard E. Smalley for their discovery in 1985 of a new form of carbon, in which the atoms are arranged in hollow spheres like this pumpkin! The new form was named Buckminsterfullerene, after the architect Buckminster Fuller who designed geodesic domes in the 1960’s.
A fullerene is any molecule composed entirely of carbon, in the form of a hollow sphere, ellipsoid or tube. Spherical fullerenes are also called buckyballs, and they resemble the balls used in soccer. Cylindrical ones are called carbon nanotubes or buckytubes.
The Chemistry and Physics of Tennis
Tennis has grown to be my favorite sport, so I found this great YouTube video of Dr. Ainissa Ramirez from Yale University explaining the science behind tennis:
Basically, modern rackets are high tech composits of carbon fibers embedded in epoxy (glue), which give them high strength and low weight. The most expensive strings are made of cow gut, but most commonly nylon is used, which is a flexible polymer.

- Nylon, a synthetic polymer used for tennis racket strings.
Strings hit the ball and create topspin, which makes the ball fall sooner than a ball without top spin. This effect is called the “Magnus effect“. This is what happens to a ball with topspin: while the ball is rotaitng, a thin layer or air around it is also rotating with it. That means that the velocity of air below it is faster than the velocity of air above it, causing it to fall sooner on the ground. That’s why you have to use top spin with your balls if you want to be too fast for your opponent.
Venus Williams usually hits a 1,200 rpm ball and Rafa Nadal 3,600 rpms. That’s faster than a revolving washing machine:-)
After reading some tennis science, I hope you agree that knowing a little bit about the chemistry and physics of tennis can really improve you as a player…! 🙂
Alcohol: Drink, Chemical, Medicine, Poison
What Is Alcohol?
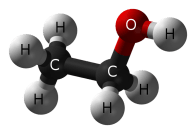
When you mention “alcohol”, you most certainly mean “ethanol”, a simple chemical which is created when grains, fruits, or vegetables are fermented. Fermentation is a process that takes place when yeast (fungi) convert food sugars into ethanol and carbon dioxide.
Sugar + yeast —> alcohol + carbon dioxide
Or
C6H12O6 + yeast ——> CH3CH2OH + CO2
Drink
 So what happens in your body when you start sipping this delicious martini????
So what happens in your body when you start sipping this delicious martini????
- Alcohol travels to your stomach.
- From the stomach, it is quickly passed into the bloodstream, where it travels in a few minutes to every part of the body, including your brain.
- In the brain alcohol affects your neurons: It alters neuron’s membranes and ion channels, enzymes such as acetylcholine, serotonin, and GABA receptors.
- When alcohol binds to these enzymes, it slows the function of the central nervous system and that’s why you feel more relaxed and stress-free.
- Ethanol also inhibits the production of a hormone (ADH) that regulates urine flow, causing increased urine production and dehydration.
- Alcohol increases dopamine by stopping enzymes that degrade it – that’s why you feel happier!
- Ethanol also causes blood vessels to dilate, resulting in flushing of the skin and a sensation of warmth as blood moves into capillaries.
- If you’ve had a large meal, alcohol will stay longer in your stomach, that’s why you can’t get drunk as easily. But eventually, it will enter the bloodstream and do its tricks:)
Your body sees alcohol as an intruder and tries to get rid of it using an enzyme to break it down, “alcohol dehydrogenase“. On contact, the enzyme snatches a hydrogen atom off the ethanol molecules in your drink, rendering it into non-intoxicating acetaldehyde, which is associated with hangovers. Alcohol dehydrogenase is less efficient in Asian populations. Lungs and liver also remove 10% of the alcohol in your body by urine and breath.
Click on this link for a schematic representation of how alcohol works in your brain.
Chemical
- Fuel: The largest single use of ethanol is as a motor fuel, fuel additive, and as a rocket fuel. Over 20% of cars in Brazil are able to run on 100% ethanol fuel.
- Raw material: Ethanol is an important industrial ingredient and has widespread use as a base chemical for other organic compounds, such as ethyl halides, ethyl esters, diethyl ether, acetic acid.
- Solvent: Ethanol is miscible with water and is a good general purpose solvent. It is found in paints, tinctures, markers, and personal care products such as perfumes and deodorants.
- Preservative: for biological specimens.
Medicine
- Antiseptic: Ethanol is used in antiseptic and some antibacterial soaps and wipes. Ethanol kills organisms by denaturing their proteins and dissolving their lipids and is effective against most bacteria and fungi, and many viruses, but is ineffective against bacterial spores.
- Solvent: As a good solvent, you will find ethanol frequently used in many medicine, such as cough syrups.
- Drug: For therapeutic neurolysis: It is injected proximate to nerve tissues and into spinal subarachnoid spaces to produce degeneration of nerve function (neurolysis) for control of chronic pain.
Poison
A couple of days ago I heard in the news that Amy Winehouse‘s death this summer was the result of alcohol poisoning….
A pathologist told a coroner’s court in north London that alcohol toxicity was the cause of the 27-year-old’s death, with her blood-alcohol levels measured at more than five times the legal limit for driving.
The report said that Amy’s blood alcohol content was 416 mg per decilitre at the time of her death. That’s 0.416% alcohol in the blood and about 9 drinks for a woman 45 kg like Amy. When large amounts of alcohol are consumed in a short period of time, alcohol poisoning can occur. Above a blood alcohol concentration of 0.5%, alcohol depresses nerves that control involuntary actions such as breathing and cardiovascular regulation. Moreover, it can cause hypothermia (low body temperature), hypoglycemia (too little blood sugar) and sever dehydration. These effects can lead to cause seizures, permanent brain damage, and ultimately death. That’s why you should never underestimate the effects of alcohol. And think twice before sipping more than three drinks in a row! Better to be safe than sorry.
How is coffee decaffeinated?
Apparently it’s National Coffee Day today, read below to learn in less than a page how coffee gets decaffeinated – not an easy task!
Coffee contains over 400 chemicals important to the taste and aroma of the final drink: it is therefore challenging to remove only caffeine while leaving the other chemicals at their original concentrations.
 To get rid of the caffeine, unroasted (green) beans are at first steamed. The beans are then rinsed with a “solvent” chemical that extracts the caffeine while leaving the other essential chemicals in the coffee beans. The process is repeated anywhere from 8 to 12 times until caffeine is removed in 97%-99.9%. So what is this magic “solvent” chemical. which is able to selectively remove caffeine while preserving the coffee aroma?
To get rid of the caffeine, unroasted (green) beans are at first steamed. The beans are then rinsed with a “solvent” chemical that extracts the caffeine while leaving the other essential chemicals in the coffee beans. The process is repeated anywhere from 8 to 12 times until caffeine is removed in 97%-99.9%. So what is this magic “solvent” chemical. which is able to selectively remove caffeine while preserving the coffee aroma?
This solvent is nothing else but carbon dioxide (CO2), which is also found in our very own atmosphere. However, as you might have guessed, CO2 is not used for decaffeination as a gas from the atmosphere. For the decaffeination process, the coffee beans are soaked in a special form of CO2 called “supercritical” CO2. OK, so now you naturally have to ask what the heck is this “supercritical” CO2??
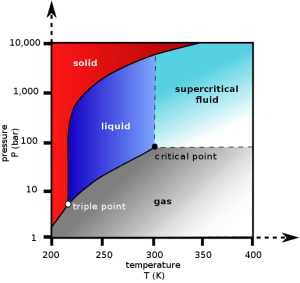
Every substance (water, hydrogen, oxygen, etc) has a so-called “critical point”. The critical point is the point above which, the distinction between liquid and gas phases of that substance stops being meaningful. That is you cannot tell any more whether this substance is gas or liquid. This new “phase”, being neither gas or liquid is called “supercritical”. A “supercritical fluid” can effuse through solids like a gas, and dissolve materials like a liquid.
So how does supercritical CO2 look like?
To achieve the supercritical state for most substances requires extreme temperatures and pressures. Carbon dioxide, however, has a fairly accessible critical point at ~ 31.1°C and 73 atm (our atmosphere has 1 atm). And it seems that supercritical CO2 has unique properties such that it can selectively extract caffeine from coffee beans, producing decaf coffee.
Carbon dioxide pressure-temperature phase diagram. The critical point is found at T=31.1°C and 73 atm. Above the critical point, one cannot really tell whether CO2 is liquid or gas. This new phase is called “supercritical”.
How does a sunblock work?
This is a question that I got very often during the summer. A sunblock can have two types of active ingredients: organic and inorganic chemicals, which reflect or scatter the light away so that it doesn’t reach the skin at all. The most common ingredients of a sunblock are zinc oxide or titanium oxide. You can usually tell who is using a sunblock just by looking, because the sunblock whites out the skin. Sort of.. like this:
Titanium dioxide (TiO2) is a white powder that has strong UV light absorbing capabilities. This property enhances TiO2 stability and enables it to protect the skin from ultraviolet light. Sunscreens designed for infants or people with sensitive skin are often based on titanium dioxide, as these mineral UV blockers are believed to cause less skin irritation than other UV absorbing chemicals. The titanium dioxide particles used in sunscreens have to be coated with silica or alumina, because titanium dioxide creates radicals in the photocatalytic reaction. These radicals are carcinogenic, and could damage the skin.
In newer formulations of sunscreens, TiO2 is combined with oil and is made up from smaller TiO2 particles (sized 40-50nm called nanoparticles). The oil in a sunscreen formula helps wet TiO2 and gives the optical effect of transparency so that you don’t have to look like someone’s painted you white.
For those who are wondering about the organic ingredients, these work primarily by absorbing UV light and dissipating it as heat. One of the most common organic molecules used in sunscreens is para-aminobenzoic acid, also know as PABA. The mechanism which allows PABA to absorb UV radiation is due to the resonance of the molecule. Resonance can be explained by saying that many molecules have more than one possible way in which the valence electrons could be placed. These electrons move freely across many nuclei. In PABA, a carbonyl group, a functional group where a carbon is double bonded to oxygen, can produce resonance by moving the valence electrons. The shift or movement of the electrons closely matches the frequency of UVB light, absorbing the light energy and releasing it as heat and or longer wavelengths of light. Source: http://www.sas.upenn.edu/~rosema/sunscreenchemistry.html
What Sunscreens/Sunblocks Screen
The portion of the sunlight that is filtered or blocked is ultraviolet radiation. There are three regions of ultraviolet light.
- UV-A penetrates deeply into the skin and can lead to DNA damage and premature skin aging.
- UV-B is involved in tanning and burning of your skin.
- UV-C, the most dangerous radiation is almost completely absorbed by the ozone in the atmosphere.
Sun is good for you, too
Although the sun can cause many aliments, UV rays are extremely important to a healthy functioning body. Vitamin D, which can not be produced by the body, is synthesized by the photolysis of a steroid in the deep epidermis of the skin. Without the exposure to natural light, the biochemical mechanism will not pursue causing disorders in both children and adults including rickets, ostomalaica, and osteoporosis.
For more info check this link.
George Whitesides on Entrepreneurship + Innovation = Jobs
The American Chemical Society (ACS) is the world’s largest scientific society with more than 161,000 members at all degree-levels and in all fields of chemistry, chemical engineering, and related fields. ACS provides a wealth of educational activities, which are mostly free and open to the public. Most importantly, they are interesting even for those who are not chemists! One of those activities is the “Virtual Career Fair”, where you can find webinars such as “Navigating the Global Industrial Job Market” and “Networking 101 — Making Your Contacts Count”. One of today’s webinars, entitled “Entrepreneurship + Innovation = Jobs” is given by Professor George Whitesides (Harvard University), a legendary innovator and pioneer, who has pioneered microfabrication and nanoscale self-assembly. One of his achievements is the “soft robot”, which is capable of gripping and lifting a raw egg without cracking its delicate shell (Angew. Chem. Int. Ed., DOI: 10.1002/anie.201006464). The challenge was to find the right material that could be soft enough to treat delicate surfaces, such as an egg. You can read the full article from C& E News here.
George Whitesides is the co-founder of a dozen companies and holds 50-plus patents. Definitely worth hearing from him about converting a great idea into a business.
From the American Chemical Society website on today’s webinar:
A recent ACS Task Force on Innovation report documented that most new jobs today and in the near future will be created by entrepreneurial start ups and small companies. Do you have an idea for a new product, service, or technology, but need help converting it into a business? Do you have a desire and the right stuff to be an entrepreneur? Plan to attend this webinar and receive valuable advice and direction from successful serial entrepreneur and Harvard University Professor George Whitesides. Whitesides recently chaired the ACS Task Force on Innovation, which was appointed by Joseph Francisco, 2010 ACS President and Professor of Chemistry at Purdue University. Whitesides and Francisco will provide valuable career advice for chemists at all stages in their careers, whether they are graduate students, postdocs, or seasoned professionals making a transition in this challenging economic job market. At this webinar, you will learn how ACS is working with U.S. policymakers, industry, academia, and its membership to support entrepreneurs and innovation to create jobs. You will learn about new ACS programs in entrepreneurship as well as specific steps that you can take now to develop the skills and find the resources needed to convert your innovative ideas into successful entrepreneurial ventures.
Enjoy.
Graphene, the strongest material on earth, now produced from cookies, roaches and dog feces
 Graphene is a material made of carbon. It is a particularly interesting material because in graphene, carbon manages to arrange itself in a sheet just one atom thick (see pic on the left). The material is so thin, that three million sheets of graphene on top of each other would be just 1mm thick. This one-atom thick sheet, densely packed in a honeycomb lattice, has excellent electrical, mechanical and thermal properties that make it the strongest material on earth, an improvement upon and a possible replacement for silicon, and the most conductive material known to man.
Graphene is a material made of carbon. It is a particularly interesting material because in graphene, carbon manages to arrange itself in a sheet just one atom thick (see pic on the left). The material is so thin, that three million sheets of graphene on top of each other would be just 1mm thick. This one-atom thick sheet, densely packed in a honeycomb lattice, has excellent electrical, mechanical and thermal properties that make it the strongest material on earth, an improvement upon and a possible replacement for silicon, and the most conductive material known to man.
In 2004, physicists at the University of Manchester and the Institute for Microelectronics Technology, Chernogolovka, Russia, first isolated individual graphene planes by exfoliating graphite (i.e. the material used for pencils) using adhesive tape. Since 2009 it has been described as the strongest material on earth, 200 times stronger than steel. In 2010, the Nobel Prize in Physics was awarded to Andre Geim and Konstantin Novoselov, “for groundbreaking experiments regarding the two-dimensional material graphene”.
It would take an elephant, balanced on a pencil, to break through a sheet of graphene the thickness of Saran Wrap.

See through: Researchers have created a flexible graphene sheet with silver electrodes printed on it (top) that can be used as a touch screen when connected to control software on a computer (bottom). Credit: Byung Hee Hong, SKKU.
said Professor James Hone of Columbia University in a statement.
As for uses? It can be used for making up new materials and electronic devices. Sort of like plastics are used nowadays but with an extra touch of technology. It could be used for transparent electronics that are stronger, cheaper, and more flexible such as shown on the right. Professor Tour of Rice University said teasingly:
You could theoretically roll up your iPhone and stick it behind your ear like a pencil.
Graphene is usually made up from graphite. But as the demand for cheap and fast large-scale graphene production becomes imminent, it quickly became clear that making graphene by splitting graphite crystals using adhesive tape, had no future.
Now a team of researchers led by Prof Tour, managed at to grow graphene directly on the backside of a copper foil at 1050°C, using six easily obtained, low or negatively valued raw carbon-containing materials used without pre-purification (cookies, chocolate, grass, plastics, roaches, and dog feces). Read the full paper here. Thanks to Anastassia for the story!
Worst comes to worst you just might end up using up that pizza from last night to get a new rollable iPhone. And think twice before you scold your dog again for doing a #2 on the carpet!