Category Archives: food
Alcohol: Drink, Chemical, Medicine, Poison
What Is Alcohol?
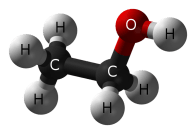
When you mention “alcohol”, you most certainly mean “ethanol”, a simple chemical which is created when grains, fruits, or vegetables are fermented. Fermentation is a process that takes place when yeast (fungi) convert food sugars into ethanol and carbon dioxide.
Sugar + yeast —> alcohol + carbon dioxide
Or
C6H12O6 + yeast ——> CH3CH2OH + CO2
Drink
 So what happens in your body when you start sipping this delicious martini????
So what happens in your body when you start sipping this delicious martini????
- Alcohol travels to your stomach.
- From the stomach, it is quickly passed into the bloodstream, where it travels in a few minutes to every part of the body, including your brain.
- In the brain alcohol affects your neurons: It alters neuron’s membranes and ion channels, enzymes such as acetylcholine, serotonin, and GABA receptors.
- When alcohol binds to these enzymes, it slows the function of the central nervous system and that’s why you feel more relaxed and stress-free.
- Ethanol also inhibits the production of a hormone (ADH) that regulates urine flow, causing increased urine production and dehydration.
- Alcohol increases dopamine by stopping enzymes that degrade it – that’s why you feel happier!
- Ethanol also causes blood vessels to dilate, resulting in flushing of the skin and a sensation of warmth as blood moves into capillaries.
- If you’ve had a large meal, alcohol will stay longer in your stomach, that’s why you can’t get drunk as easily. But eventually, it will enter the bloodstream and do its tricks:)
Your body sees alcohol as an intruder and tries to get rid of it using an enzyme to break it down, “alcohol dehydrogenase“. On contact, the enzyme snatches a hydrogen atom off the ethanol molecules in your drink, rendering it into non-intoxicating acetaldehyde, which is associated with hangovers. Alcohol dehydrogenase is less efficient in Asian populations. Lungs and liver also remove 10% of the alcohol in your body by urine and breath.
Click on this link for a schematic representation of how alcohol works in your brain.
Chemical
- Fuel: The largest single use of ethanol is as a motor fuel, fuel additive, and as a rocket fuel. Over 20% of cars in Brazil are able to run on 100% ethanol fuel.
- Raw material: Ethanol is an important industrial ingredient and has widespread use as a base chemical for other organic compounds, such as ethyl halides, ethyl esters, diethyl ether, acetic acid.
- Solvent: Ethanol is miscible with water and is a good general purpose solvent. It is found in paints, tinctures, markers, and personal care products such as perfumes and deodorants.
- Preservative: for biological specimens.
Medicine
- Antiseptic: Ethanol is used in antiseptic and some antibacterial soaps and wipes. Ethanol kills organisms by denaturing their proteins and dissolving their lipids and is effective against most bacteria and fungi, and many viruses, but is ineffective against bacterial spores.
- Solvent: As a good solvent, you will find ethanol frequently used in many medicine, such as cough syrups.
- Drug: For therapeutic neurolysis: It is injected proximate to nerve tissues and into spinal subarachnoid spaces to produce degeneration of nerve function (neurolysis) for control of chronic pain.
Poison
A couple of days ago I heard in the news that Amy Winehouse‘s death this summer was the result of alcohol poisoning….
A pathologist told a coroner’s court in north London that alcohol toxicity was the cause of the 27-year-old’s death, with her blood-alcohol levels measured at more than five times the legal limit for driving.
The report said that Amy’s blood alcohol content was 416 mg per decilitre at the time of her death. That’s 0.416% alcohol in the blood and about 9 drinks for a woman 45 kg like Amy. When large amounts of alcohol are consumed in a short period of time, alcohol poisoning can occur. Above a blood alcohol concentration of 0.5%, alcohol depresses nerves that control involuntary actions such as breathing and cardiovascular regulation. Moreover, it can cause hypothermia (low body temperature), hypoglycemia (too little blood sugar) and sever dehydration. These effects can lead to cause seizures, permanent brain damage, and ultimately death. That’s why you should never underestimate the effects of alcohol. And think twice before sipping more than three drinks in a row! Better to be safe than sorry.
How is coffee decaffeinated?
Apparently it’s National Coffee Day today, read below to learn in less than a page how coffee gets decaffeinated – not an easy task!
Coffee contains over 400 chemicals important to the taste and aroma of the final drink: it is therefore challenging to remove only caffeine while leaving the other chemicals at their original concentrations.
 To get rid of the caffeine, unroasted (green) beans are at first steamed. The beans are then rinsed with a “solvent” chemical that extracts the caffeine while leaving the other essential chemicals in the coffee beans. The process is repeated anywhere from 8 to 12 times until caffeine is removed in 97%-99.9%. So what is this magic “solvent” chemical. which is able to selectively remove caffeine while preserving the coffee aroma?
To get rid of the caffeine, unroasted (green) beans are at first steamed. The beans are then rinsed with a “solvent” chemical that extracts the caffeine while leaving the other essential chemicals in the coffee beans. The process is repeated anywhere from 8 to 12 times until caffeine is removed in 97%-99.9%. So what is this magic “solvent” chemical. which is able to selectively remove caffeine while preserving the coffee aroma?
This solvent is nothing else but carbon dioxide (CO2), which is also found in our very own atmosphere. However, as you might have guessed, CO2 is not used for decaffeination as a gas from the atmosphere. For the decaffeination process, the coffee beans are soaked in a special form of CO2 called “supercritical” CO2. OK, so now you naturally have to ask what the heck is this “supercritical” CO2??
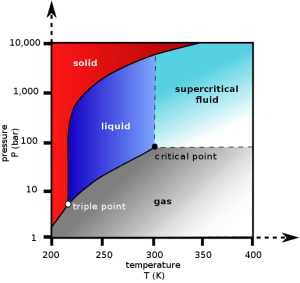
Every substance (water, hydrogen, oxygen, etc) has a so-called “critical point”. The critical point is the point above which, the distinction between liquid and gas phases of that substance stops being meaningful. That is you cannot tell any more whether this substance is gas or liquid. This new “phase”, being neither gas or liquid is called “supercritical”. A “supercritical fluid” can effuse through solids like a gas, and dissolve materials like a liquid.
So how does supercritical CO2 look like?
To achieve the supercritical state for most substances requires extreme temperatures and pressures. Carbon dioxide, however, has a fairly accessible critical point at ~ 31.1°C and 73 atm (our atmosphere has 1 atm). And it seems that supercritical CO2 has unique properties such that it can selectively extract caffeine from coffee beans, producing decaf coffee.
Carbon dioxide pressure-temperature phase diagram. The critical point is found at T=31.1°C and 73 atm. Above the critical point, one cannot really tell whether CO2 is liquid or gas. This new phase is called “supercritical”.