Category Archives: Uncategorized
EU and Me
 Last week I had the honour to be interviewed by the European Commission by Eleni-Revekka Staiou and Jef Ponsaers for EU and Me, a digital story campaign showing that living in the European Union provides endless opportunities to do what you love – wherever that may lead. EU can help unlock your potential and ignite your inspiration giving you the tools to pursue your passion!
Last week I had the honour to be interviewed by the European Commission by Eleni-Revekka Staiou and Jef Ponsaers for EU and Me, a digital story campaign showing that living in the European Union provides endless opportunities to do what you love – wherever that may lead. EU can help unlock your potential and ignite your inspiration giving you the tools to pursue your passion!
How did EU helped my research as a Chemist?
When i came back to Greece from the US, I received the Marie Curie Fellowship, that helped me setup my lab, hire my first lab staff, produce independent publications, travel to conferences to build up my own research network. I could invite my colleagues from the US, and the fellowship also paid for overheads in my home institute, the Biomedical Research Foundation of the Academy of Athens.
 The National Strategic Reference Program co-funded by the EU (ESPA in Greek) awarded more funds to my lab to continue our research (following open call contests).
The National Strategic Reference Program co-funded by the EU (ESPA in Greek) awarded more funds to my lab to continue our research (following open call contests).
PRACE (Partnership for Advanced Computing in Europe), provided computational time to run our demanding molecular simulations for discovering new drugs (over 45M core hours).
Through European Union’s main funding scheme, Horizon 2020, I am implicated in projects like VI-SEEM, which has built an e-infrastructure platform for Southeastern Europe & Eastern Mediterranean, and also the Human Brain Project through which we work to discover new treatments for brain diseases!
Through awards I won, such as PRACE Ada Lovelace 2016, I could reach out to citizens (and especially girls), get them acquainted with science & tell them how research leads to products that we use in our everyday life.
Finally EU through its many initiatives helped to host & collaborate with many students from across Europe!
To name a few: HPC-Europa3, PRACE Summer of HPC, ESPA, ERASMUS+.
The NEXT STEP? The European Bank for Reconstruction & Development has now signed an agreement to build a new Centre for Personalized Medicine in my home institute, the Biomedical Research Foundation.
All that has led & continues to lead to new candidate drugs that we hope in the future will improve the quality of life for thousands of citizens in the world!
The opportunities are out there for you too – Go grab them!
PS. if you are wondering why I am extending my hand like this in the first picture – this was supposed to be a selfie video 🙂
Free Online 2015 Drug Design and Delivery Symposium by ACS
The American Chemical Society oranizes a free four-module symposiumon Drug Design and Delivery.
The four modules contain multiple talks on the drug discovery process and are delivered by expert chemists working in top pharmaceutical companies in the world. Attendants will learn about Strategies to Improve Solubility of Drug Candidates, PAINS filters, using PET for Labeling in Drug Discovery & Development, Pharmacokinetic Considerations in Drug Design and Development, and more.
Module 1: Improving Drug Design Efficiency and Efficacy
Module 2: Activity/Potency Screening for Drug Lead and Candidate Optimization
Module 3: Enabling Drug Delivery
Module 4: Pharmacokinetics
Enjoy!
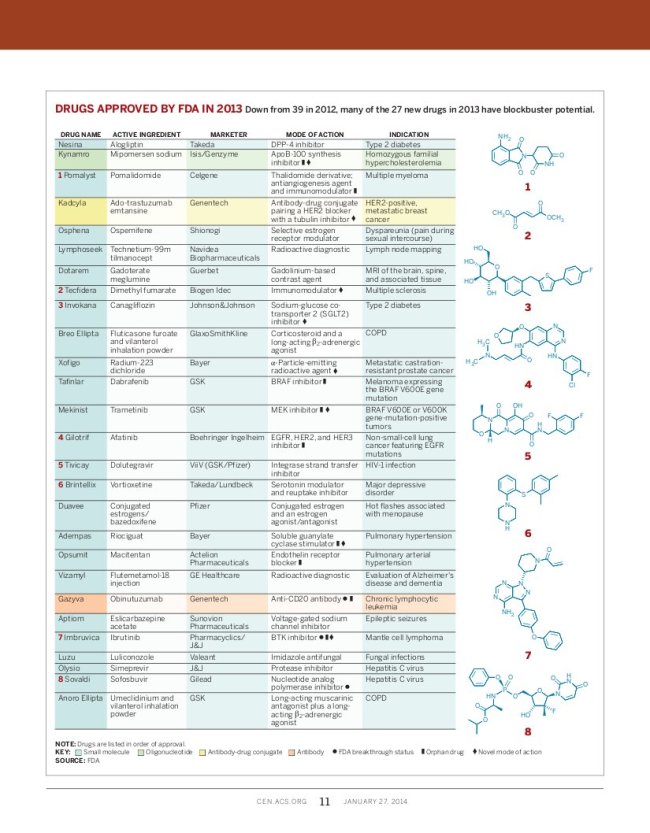
FDA Approves 27 New Drugs in 2013
The new drug approvals for 2013 were mostly cancer drugs and drugs for orphan diseases.
Other therapeutic areas included metabolic and endocrinological therapies (two type 2 diabetes drugs and one dyslipidaemia drug), antivirals (two hepatitis C virus (HCV) drugs and an HIV drug) and medical imaging products (three approvals).
Highlights
Several of these new drugs are scientifically novel.
Gilead’s sofosbuvir is first-in-class NS5B polymerase inhibitor and the first oral HCV drug (Nature Rev. Drug Discov. 12, 409–411; 2013).
Pharmacyclics and Janssen’s ibrutinib is a first-in-class Bruton’s tyrosine kinase (BTK) inhibitor, which entered the clinic in 2009, and is now approved for an aggressive form of B cell non-Hodgkin’s lymphoma, but it is now being also tested for other types of cancers such as chronic lymphocytic leukaemia (CLL). Ibrutinib is the second irreversible kinase inhibitor to be approved after afatinib, setting new hope for the development of irreversible agents that have been thought difficult to reach the market in earlier years (Nature Rev. Drug Discov. 12, 649–651; 2013).
Biogen Idec’s dimethyl fumarate (formerly BG-12) is an extremely small molecule (MW only 144 Da) that had been approved as an anti-psoriasis drug in Germany as well as for preventing the growth of mold in furniture! Now Biogen Idec gets its approval for dimethyl fumarate as the third oral drug for multiple sclerosis. Drug mechanism remains unclear.
GSK’s trametinib is a first-in-class MAPK/ERK kinase (MEK) inhibitor that overcame bioavailability, pharmacokinetics and toxicity problems of previous drug candidates (Nature Rev. Drug Discov. 11, 819–820; 2012). Trametinib is approved for cancers bearing specific BRAF mutations (such as metastatic melanoma). GSK picked up five approvals from the FDA last year (counting the approval granted to ViiV), more than any other drug developer in 2013.
Janssen’s canagliflozin, is the first-FDA approved sodium-dependent glucose cotransporter 2 (SGLT2) inhibitor, against type 2 diabetes space.
Genzyme and Isis’s mipomersen acts on a novel target, binding apolipoprotein B mRNA, reducing protein synthesis and thereby lowering low-density lipoprotein (LDL) levels. Because of its side effects it may be used only as an orphan disease drug agains homozygous familial hypercholesterolaemia and not as a general cholesterol-lowering agent. Regulators in the European Union (EU) have rejected mipomersen because of its side-effects.
Sources:
Industry – academia partnerships: way to go for anticancer drug development?
Industry – academia partnerships are increasingly growing in an effort to make the drug discovery process more efficient. In a Cancer Research UK perspective, several industrial collaborations are outlined with an eye on the distinct but com-plementary expertise brought about in anti-cancer drug development.
For more industry – academia initiatives check out
GlaxoSmithKline’s Discovery Fast Track competition for academic researchers
Eli Lilly’s Open Innovation Platform
Marie Curie’s Industry-Academia Partnerships and Pathways (IAPP)
Read another article on industry – academia partnerships from Science Magazine and an older article from this blog on the topic here.
2012 in review
The WordPress.com stats helper monkeys prepared a 2012 annual report for Life is Chemistry.
Here’s an excerpt:
600 people reached the top of Mt. Everest in 2012. This blog got about 4,900 views in 2012. If every person who reached the top of Mt. Everest viewed this blog, it would have taken 8 years to get that many views.
The Nobel Prize in Physics 2012… explained
The Nobel prize in Physics 2012 was jointly awarded yesterday to Serge Haroche and David J. Wineland “for ground-breaking experimental methods that enable measuring and manipulation of individual quantum systems”.
Haroche and Wineland showed with real experiments of photons that it is possible for a quantum system to exist in two states at the same time. Originally, this was proposed by the Austrian physicist Erwin Schroedinger (Nobel Prize in Physics, 1933) within the quantum theory framework. The fact that a photon or an atom can exist in two states at the same time can have practical applications in life, i.e. in the quantum computer. Today’s computers store information in bits, which can have the value of 0 or 1 only. In quantum computing, a bit can exist in 0 and 1 at the same time (qubit), allowing for parallel computations at astronomical speeds. The quantum computer might revolutionize our lives the way computer have changed the way we live in the past century.
Back to my alma mater
Yesterday, I travelled to my alma mater, Universitaet Heidelberg, to be part of the celebrations for the University’s 625th anniversary. When I arrived in Heidelberg, I realized that I was celebrating my own anniversary as well: ten years in science. Ten years since I first landed in Heidelberg to start my doctoral studies. These years in science and my Doktorvater’s blog inspired me to start my own blog today and write about my life and the science which is everywhere: chemistry.