Category Archives: science facts
366 Days: The Year in Science
Read below the Science Review of 2012 by Nature Magazine, with Greece making it to the top 22 “leading science nations” with 1% of the ‘most cited papers’!
Also, read on Science Magazine’s Breakthrough of the Year 2012 (Higgs Boson) and the runners-up: Genome Engineering, Curiosity Landing, Bionics, Eggs from Stem Cells, Encode, X-ray laser advances and more!
http://www.sciencemag.org/site/special/btoy2012/
Alcohol: Drink, Chemical, Medicine, Poison
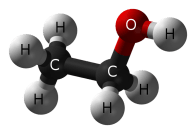
What Is Alcohol?
When you mention “alcohol”, you most certainly mean “ethanol”, a simple chemical which is created when grains, fruits, or vegetables are fermented. Fermentation is a process that takes place when yeast (fungi) convert food sugars into ethanol and carbon dioxide.
Sugar + yeast —> alcohol + carbon dioxide
Or
C6H12O6 + yeast ——> CH3CH2OH + CO2
Drink
 So what happens in your body when you start sipping this delicious martini????
So what happens in your body when you start sipping this delicious martini????
- Alcohol travels to your stomach.
- From the stomach, it is quickly passed into the bloodstream, where it travels in a few minutes to every part of the body, including your brain.
- In the brain alcohol affects your neurons: It alters neuron’s membranes and ion channels, enzymes such as acetylcholine, serotonin, and GABA receptors.
- When alcohol binds to these enzymes, it slows the function of the central nervous system and that’s why you feel more relaxed and stress-free.
- Ethanol also inhibits the production of a hormone (ADH) that regulates urine flow, causing increased urine production and dehydration.
- Alcohol increases dopamine by stopping enzymes that degrade it – that’s why you feel happier!
- Ethanol also causes blood vessels to dilate, resulting in flushing of the skin and a sensation of warmth as blood moves into capillaries.
- If you’ve had a large meal, alcohol will stay longer in your stomach, that’s why you can’t get drunk as easily. But eventually, it will enter the bloodstream and do its tricks:)
Your body sees alcohol as an intruder and tries to get rid of it using an enzyme to break it down, “alcohol dehydrogenase“. On contact, the enzyme snatches a hydrogen atom off the ethanol molecules in your drink, rendering it into non-intoxicating acetaldehyde, which is associated with hangovers. Alcohol dehydrogenase is less efficient in Asian populations. Lungs and liver also remove 10% of the alcohol in your body by urine and breath.
Click on this link for a schematic representation of how alcohol works in your brain.
Chemical
- Fuel: The largest single use of ethanol is as a motor fuel, fuel additive, and as a rocket fuel. Over 20% of cars in Brazil are able to run on 100% ethanol fuel.
- Raw material: Ethanol is an important industrial ingredient and has widespread use as a base chemical for other organic compounds, such as ethyl halides, ethyl esters, diethyl ether, acetic acid.
- Solvent: Ethanol is miscible with water and is a good general purpose solvent. It is found in paints, tinctures, markers, and personal care products such as perfumes and deodorants.
- Preservative: for biological specimens.
Medicine
- Antiseptic: Ethanol is used in antiseptic and some antibacterial soaps and wipes. Ethanol kills organisms by denaturing their proteins and dissolving their lipids and is effective against most bacteria and fungi, and many viruses, but is ineffective against bacterial spores.
- Solvent: As a good solvent, you will find ethanol frequently used in many medicine, such as cough syrups.
- Drug: For therapeutic neurolysis: It is injected proximate to nerve tissues and into spinal subarachnoid spaces to produce degeneration of nerve function (neurolysis) for control of chronic pain.
Poison
A couple of days ago I heard in the news that Amy Winehouse‘s death this summer was the result of alcohol poisoning….
A pathologist told a coroner’s court in north London that alcohol toxicity was the cause of the 27-year-old’s death, with her blood-alcohol levels measured at more than five times the legal limit for driving.
The report said that Amy’s blood alcohol content was 416 mg per decilitre at the time of her death. That’s 0.416% alcohol in the blood and about 9 drinks for a woman 45 kg like Amy. When large amounts of alcohol are consumed in a short period of time, alcohol poisoning can occur. Above a blood alcohol concentration of 0.5%, alcohol depresses nerves that control involuntary actions such as breathing and cardiovascular regulation. Moreover, it can cause hypothermia (low body temperature), hypoglycemia (too little blood sugar) and sever dehydration. These effects can lead to cause seizures, permanent brain damage, and ultimately death. That’s why you should never underestimate the effects of alcohol. And think twice before sipping more than three drinks in a row! Better to be safe than sorry.
What are we really made of?
Watch this!! – I just found this amazing video that offers a very entertaining introduction into the particles and forces that make up me, you, the earth, the universe. Featuring Morgan Freeman, Stephen Hawking, Michio Kaku, Brian Cox, Richard Feynman, and Frank Close, this video offers a cursory introduction to quantum chemistry – the science that governs the interactions of subatomic particles, atoms, and even molecules.
Basically, in 3 and a half minutes, the video tries to explain that:
Dig deep inside the atom
and you’ll find tiny particles
Held together by invisible forces
The atoms that we’re made of have
Negatively charged electrons
Whirling around a big bulky nucleus
The universe is made of
Twelve particles of matter
Four forces of nature
The world is a dynamic mess
Of jiggling things
In the quantum world electrons behave as waves and as particles
For the ultimate triumph of science
We need a theory of everything
Which is still just beyond our grasp
The theory of everything is the Holy Grail of Physics: A theory that will explain through a single model everything from Creation, to supernovas, to atoms and molecules, perhaps even DNA, people, and love. (The four yet unified forces of nature are: gravitation, electromagnetism and the weak and strong nuclear forces that keep together elementary particles.)
If we ever create this theory, we might answer questions such as:
- What happened before the big bang?
- Is it possible to build a time machine?
- Can we punch a hole in space?
“The Quantum World” music video is the eleventh installment in the ongoing Symphony of Science music video series. Materials used in the creation of this video are from:
http://symphonyofscience.com for downloads & more videos!
Why is the sky blue?
A question that I get very often is why did I ever return to Greece to do science. It is indeed hard to explain why would a young scientist leave an educational and technological paradise such as Yale University to seek a better future in a country on the verge of default. So, I usually start by explaining the ties to my family, my friends, the ease to speak my own language, etc. But then again only one picture could actually summarize it 🙂
In a country with such blue skies, I often get bombarded with questions from my non-scientist friends such as: “Why is the sky blue? Why is the sunset red?”. So here it is in very simple words:
Light
Light looks white, but it is actually a combination of colors (you’ve all see a rainbow right?). These colors are in order: red, orange, yellow, green, blue, indigo and violet. These colors all have different energies, violet has the highest energy and red the lowest. Light travels in a straight line when it is undisturbed, such as in space. What happens when light enters the atmosphere?
Atmoshpere – Blue Sky
The atmosphere is composed by particles such as dust and water that can be seen with the naked eye, but also smaller particles such as oxygen and nitrogen that are so small that we cannot see them. When light enters the atmosphere it hits these small molecules such as oxygen (21%) and nitrogen (78%) (gases). Gases then tend to absorb not all of the light but only part of it. By “part of the light” I mean that they absorb only one out of 7 colors. It so happens that this color is blue. After a while, the gas molecules give off this blue light in all different directions in the atmosphere. The blue light is scattered everywhere you look in the atmosphere. This is why the sky is blue. This is also why the sun looks yellow (remember light is white). Because if you take off blue from the rainbow, all the remaining colors together look yellow.
Atmosphere – Red Sunset
So why isn’t the sky blue also at sunset? At sunset, the sun is close to the horizon and light needs to pass through a much longer path through the atmosphere to reach you than at noon. Close to the horizon different particles (such as aerosols) are concentrated. It so happens that these aerosols absorb the red light instead of the blue for a while and then give off this red light at all possible directions such as oxygen and nitrogen did for the blue light. Therefore we can enjoy red sunsets and blue skies…