Monthly Archives: November 2011
Alcohol: Drink, Chemical, Medicine, Poison
What Is Alcohol?
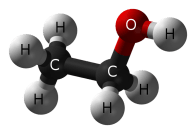
When you mention “alcohol”, you most certainly mean “ethanol”, a simple chemical which is created when grains, fruits, or vegetables are fermented. Fermentation is a process that takes place when yeast (fungi) convert food sugars into ethanol and carbon dioxide.
Sugar + yeast —> alcohol + carbon dioxide
Or
C6H12O6 + yeast ——> CH3CH2OH + CO2
Drink
 So what happens in your body when you start sipping this delicious martini????
So what happens in your body when you start sipping this delicious martini????
- Alcohol travels to your stomach.
- From the stomach, it is quickly passed into the bloodstream, where it travels in a few minutes to every part of the body, including your brain.
- In the brain alcohol affects your neurons: It alters neuron’s membranes and ion channels, enzymes such as acetylcholine, serotonin, and GABA receptors.
- When alcohol binds to these enzymes, it slows the function of the central nervous system and that’s why you feel more relaxed and stress-free.
- Ethanol also inhibits the production of a hormone (ADH) that regulates urine flow, causing increased urine production and dehydration.
- Alcohol increases dopamine by stopping enzymes that degrade it – that’s why you feel happier!
- Ethanol also causes blood vessels to dilate, resulting in flushing of the skin and a sensation of warmth as blood moves into capillaries.
- If you’ve had a large meal, alcohol will stay longer in your stomach, that’s why you can’t get drunk as easily. But eventually, it will enter the bloodstream and do its tricks:)
Your body sees alcohol as an intruder and tries to get rid of it using an enzyme to break it down, “alcohol dehydrogenase“. On contact, the enzyme snatches a hydrogen atom off the ethanol molecules in your drink, rendering it into non-intoxicating acetaldehyde, which is associated with hangovers. Alcohol dehydrogenase is less efficient in Asian populations. Lungs and liver also remove 10% of the alcohol in your body by urine and breath.
Click on this link for a schematic representation of how alcohol works in your brain.
Chemical
- Fuel: The largest single use of ethanol is as a motor fuel, fuel additive, and as a rocket fuel. Over 20% of cars in Brazil are able to run on 100% ethanol fuel.
- Raw material: Ethanol is an important industrial ingredient and has widespread use as a base chemical for other organic compounds, such as ethyl halides, ethyl esters, diethyl ether, acetic acid.
- Solvent: Ethanol is miscible with water and is a good general purpose solvent. It is found in paints, tinctures, markers, and personal care products such as perfumes and deodorants.
- Preservative: for biological specimens.
Medicine
- Antiseptic: Ethanol is used in antiseptic and some antibacterial soaps and wipes. Ethanol kills organisms by denaturing their proteins and dissolving their lipids and is effective against most bacteria and fungi, and many viruses, but is ineffective against bacterial spores.
- Solvent: As a good solvent, you will find ethanol frequently used in many medicine, such as cough syrups.
- Drug: For therapeutic neurolysis: It is injected proximate to nerve tissues and into spinal subarachnoid spaces to produce degeneration of nerve function (neurolysis) for control of chronic pain.
Poison
A couple of days ago I heard in the news that Amy Winehouse‘s death this summer was the result of alcohol poisoning….
A pathologist told a coroner’s court in north London that alcohol toxicity was the cause of the 27-year-old’s death, with her blood-alcohol levels measured at more than five times the legal limit for driving.
The report said that Amy’s blood alcohol content was 416 mg per decilitre at the time of her death. That’s 0.416% alcohol in the blood and about 9 drinks for a woman 45 kg like Amy. When large amounts of alcohol are consumed in a short period of time, alcohol poisoning can occur. Above a blood alcohol concentration of 0.5%, alcohol depresses nerves that control involuntary actions such as breathing and cardiovascular regulation. Moreover, it can cause hypothermia (low body temperature), hypoglycemia (too little blood sugar) and sever dehydration. These effects can lead to cause seizures, permanent brain damage, and ultimately death. That’s why you should never underestimate the effects of alcohol. And think twice before sipping more than three drinks in a row! Better to be safe than sorry.
Celebrating Marie Curie
7 November 2011 is a special day for Chemistry. It marks the 144th anniversary of Marie Curie’s birthday; 2011 has been designated as the International Year of Chemistry (IYC) by IUPAC and UNESC.
And there’s even more: The IYC 2011 coincides with the 100th anniversary of Marie Curie’s Nobel Prize in Chemistry. France and Poland declared 2011 to be the Year of Marie Curie. To recognise her achievements, Marie Curie has been chosen as the symbol for this year’s 2011 IYC – to celebrate it Google has dedicated today its search page to Marie Curie.
Marie Curie is shown by Google at her work bench – indeed she is known for her vigorous passion for science, her hard work that led her to claim two Nobel prizes, and her enormous contributions to Chemistry and the fight against cancer. Her legacy is lived on in several academic institutions and charities such as the Marie Curie Cancer Care. Below are important facts about the “most inspirational woman in science”:
- Marie Sklodowska Curie was born on 7 November 1867 in Warsaw.
- She was awarded the 1903 Nobel prize in Physics, together with husband Pierre Curie and Henri Becquerel for her “researches on the radiation phenomena”.
- On 8 Nov. 1911, she was awarded the 1911 Nobel prize in Chemistry for “the discovery of radium and polonium, the isolation of radium and the study of radium’s nature and compounds”.
- In 1906 she became the first female Sorbonne Professor.
- She is the first of only two people ever to win the Nobel prize in multiple fields. She was also the first woman ever to win the Nobel Prize.
- Curie promoted the use of the radioactivity for therapeutic purposes.
- In 1914 she helped develop small, mobile X-ray units and joined the war front with her 17-year old daughter Irene to help wounded soldierslocate fractures, bullets, and shrapnel.
- Her daughter Irene won the 1935 chemistry Nobel for her work on artificial radioactivity.
- She founded the Curie Institutes in France and Poland, co-founded the Warsaw Radium Institute, and headed the Pasteur Institute.
Curie helped forever change how the world perceived women in science and set a shining example for the future generations of scientists in that rigorous and determined investigation can lead to remarkable discoveries.
7 November is also a very special day for me as it is also my own birthday. I am unbelievably honored to share a birthday with a female chemist of this caliber. I am continuously inspired by her lifelong dedication and contributions to science. As she very well put it:
Nothing in life is to be feared, it is only to be understood. Now is the time to understand more, so that we may fear less.
–Marie Curie
The biography of Marie Curie was written by her daughter, Eve Curie. From Amazon.com:
From Amazon.com:
This biography chronicles Curie’s legendary achievements in science, including her pioneering efforts in the study of radioactivity and her two Nobel Prizes in Physics and Chemistry. It also spotlights her remarkable life, from her childhood in Poland, to her storybook Parisian marriage to fellow scientist Pierre Curie, to her tragic death from the very radium that brought her fame.
Below are some of the many events with which the IYC2011 has honored Marie Curie:
- A Marie Curie inspired poster exhibition was held at the Research Centre for Materials Science, Nagoya University (July 21st -Aug 31st 2011) in collaboration with The Curie Museum and Curie Institute.
- To celebrate her achievements, a re-enactment play inspired by the life of Marie Curie was performed at the IUPAC World Chemistry Congress in San Juan Puerto Rico by professional actress, Susan Frontczak, which was followed by the award ceremony for the 23 Distinguished Women in Chemistry/Chemical Engineering (August 2nd 2011)
- The Geffen Playhouse in Los Angeles, CA is producing the world premiere of Radiance: The Passion of Marie Curie (Nov.1 through Dec. 11, 2011).
- Marie Curie on Stamps Exhibition @ Postalia, Québec city
- 25 November 2011- MSC-100 closing celebration to be held at the Royal Castle in Warsaw.