ChemMatters: Demystifying Everyday Chemistry for high school students
ChemMatters is a magazine for high school students and high school teachers published by the Education Division of the American Chemical Society.
The magazine, which appears four times a year, contains articles that feature real-world applications of chemistry concepts introduced in the classroom. Latest issues cover themes such as the application of nanotechnology, great discoveries in chemistry, and the science behind weather folklore.
The site provides a Teacher’s guide, videos, material for the classroom and more.
Check out the ChemMatters web site for the latest issue!
View the lastest Episode on ChemMatters: “Episode 10: Graphene: The Next Wonder Material?” on carbon allotropes and the applications of graphene.
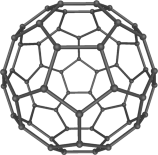
Happy Halloween! (with a fullerene twist)
The 1996 Nobel Prize for Chemistry has been won by Harold W. Kroto, Robert F. Curl and Richard E. Smalley for their discovery in 1985 of a new form of carbon, in which the atoms are arranged in hollow spheres like this pumpkin! The new form was named Buckminsterfullerene, after the architect Buckminster Fuller who designed geodesic domes in the 1960’s.
A fullerene is any molecule composed entirely of carbon, in the form of a hollow sphere, ellipsoid or tube. Spherical fullerenes are also called buckyballs, and they resemble the balls used in soccer. Cylindrical ones are called carbon nanotubes or buckytubes.
The Nobel Prize in Physics 2012… explained
The Nobel prize in Physics 2012 was jointly awarded yesterday to Serge Haroche and David J. Wineland “for ground-breaking experimental methods that enable measuring and manipulation of individual quantum systems”.
Haroche and Wineland showed with real experiments of photons that it is possible for a quantum system to exist in two states at the same time. Originally, this was proposed by the Austrian physicist Erwin Schroedinger (Nobel Prize in Physics, 1933) within the quantum theory framework. The fact that a photon or an atom can exist in two states at the same time can have practical applications in life, i.e. in the quantum computer. Today’s computers store information in bits, which can have the value of 0 or 1 only. In quantum computing, a bit can exist in 0 and 1 at the same time (qubit), allowing for parallel computations at astronomical speeds. The quantum computer might revolutionize our lives the way computer have changed the way we live in the past century.
The Chemistry and Physics of Tennis
Tennis has grown to be my favorite sport, so I found this great YouTube video of Dr. Ainissa Ramirez from Yale University explaining the science behind tennis:
Basically, modern rackets are high tech composits of carbon fibers embedded in epoxy (glue), which give them high strength and low weight. The most expensive strings are made of cow gut, but most commonly nylon is used, which is a flexible polymer.

- Nylon, a synthetic polymer used for tennis racket strings.
Strings hit the ball and create topspin, which makes the ball fall sooner than a ball without top spin. This effect is called the “Magnus effect“. This is what happens to a ball with topspin: while the ball is rotaitng, a thin layer or air around it is also rotating with it. That means that the velocity of air below it is faster than the velocity of air above it, causing it to fall sooner on the ground. That’s why you have to use top spin with your balls if you want to be too fast for your opponent.
Venus Williams usually hits a 1,200 rpm ball and Rafa Nadal 3,600 rpms. That’s faster than a revolving washing machine:-)
After reading some tennis science, I hope you agree that knowing a little bit about the chemistry and physics of tennis can really improve you as a player…! 🙂
The Chemistry of Love
I was sitting in the lobby of my accountant’s office, flipping absentmindedly through a magazine when she walked in. I’ve never had a visceral reaction as when I saw her walk through that door. There was just something about her; I felt head over heels… My heart started racing and I had butterflies in my stomach…
 This is the amazing time when you are truly love-struck. With an irresistible cocktail of chemicals, our brain entices us to fall in love. But is it really us or is it yet another nature’s trick to keep our species alive?
This is the amazing time when you are truly love-struck. With an irresistible cocktail of chemicals, our brain entices us to fall in love. But is it really us or is it yet another nature’s trick to keep our species alive?
Scientists agree that there are three stages and processes in love:
Stage 1 – Attraction : Dopamine and Adrenaline
: Dopamine and Adrenaline
When you fall in love, your brain starts sending signals before you can even blink. Your heart races and palms sweat: adrenaline is getting released from neurons. Then, when you are close to your sweetheart, dopamine is released, which triggers euphoria and feelings of bliss, increased energy, increased energy, less need for sleep or food, and focused attention on your new relationship. Phenylethylamine (PEA) is the chemical responsible for releasing adrenaline and dopamine. Biologically speaking, with the combination of dopamine, adrenaline, and PEA in your body, you’re experiencing something similar to a cocaine high. And it doesn’t matter whether you just met this *special* person or you ‘ve known them for ages; these love signals can be released at any time. Actually, no, not at any time. Only when mother nature thinks the timing is right** (see below).
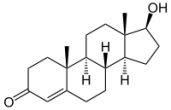
Stage 2 – Lust: Testosterone kicks in
That is about the time that androgens and estrogens kick in. Estrogens and androgens are steroids, which are the female and male sex hormones, respectively. Testosterone is the sex hormone in both male and females, which is what gives us libido and sex drive. This hormone is elevated when we are in love. This makes sense as you’re most concerned about sex when it’s most likely to result in reproduction. Some people argue that this Stage precedes Stage 1, but it’s up to you to decide 🙂
Stage 3 – Attachment: Oxytocin, the love hormone
In the long run, two other hormones make their headway when emotionally involved: the “hormones of attachment”, oxytocin and vasopressin.
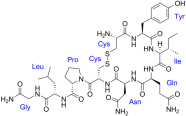
Oxytocin. Notice something? As we go down the love chain, the molecules appear more complex; sort of like love-life!
Oxytocin is a hormone stored in the brain. When the hormone is released in the brain, it results in feelings of attachment and bonding. It has been found that oxytocin gets released at orgasm, which is why couples feel much closer to one another after they have had sex. Oxytocin is also released at childbirth and is partly responsible for the strong bond between mum and baby (actually oxytocin has a Greek root: “οξυτοκίνη”, which means “quick birth”). So, if you feel the urge for a stay-at-home movie night to cuddle with your beau, it may be oxytocin at work.
Vasopressin is another important hormone when it comes to relationships. Scientists studied the prairie vole, which is known to be monogamous and to express vasopressin like humans. When the researchers introduced vasopressin receptors to the “meadow vole”, the promiscuous cousin of the prairie vole, these playboys reformed their ways and fixated on one female, choosing to mate with only her – even when other females tried to tempt them!!
Wondering why some people find it hard to get emotionally involved or committed?
Some people might be less prone to form stable relationships because they might be not express as many “oxytocin or vasopressin receptors” (proteins that take up oxytocin or vasopressin) in their brains. Thus, some people are not that sensitive to the “commitment hormones” oxytocin and vasopressin. Experiments in animals showed that if you remove the oxytocin or vasopressin receptors, these animals cannot form strong bonds with each other any more.
Timing is important. The perfect partner can sit right next to you at a party, and you might not notice him or her if you’re too busy at work, enmeshed in another relationship, or otherwise preoccupied. But if you’ve just moved to a new city, recovered from an unsatisfying love affair, begun to make enough money to raise a family, are suffering through a difficult experience, or have a good deal of spare time, you are ripe to fall in love.
So love appears to all be yet another one of nature’s traits:
Romantic love evolved to enable you to focus your energy on just one person at a time, conserving time and energy. The sex drive evolved to get you out there looking for partners. Attachment, the feeling of security you can feel with a long-term partner, evolved to help you stay together long enough to raise kids.
Alcohol: Drink, Chemical, Medicine, Poison
What Is Alcohol?
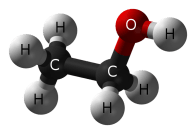
When you mention “alcohol”, you most certainly mean “ethanol”, a simple chemical which is created when grains, fruits, or vegetables are fermented. Fermentation is a process that takes place when yeast (fungi) convert food sugars into ethanol and carbon dioxide.
Sugar + yeast —> alcohol + carbon dioxide
Or
C6H12O6 + yeast ——> CH3CH2OH + CO2
Drink
 So what happens in your body when you start sipping this delicious martini????
So what happens in your body when you start sipping this delicious martini????
- Alcohol travels to your stomach.
- From the stomach, it is quickly passed into the bloodstream, where it travels in a few minutes to every part of the body, including your brain.
- In the brain alcohol affects your neurons: It alters neuron’s membranes and ion channels, enzymes such as acetylcholine, serotonin, and GABA receptors.
- When alcohol binds to these enzymes, it slows the function of the central nervous system and that’s why you feel more relaxed and stress-free.
- Ethanol also inhibits the production of a hormone (ADH) that regulates urine flow, causing increased urine production and dehydration.
- Alcohol increases dopamine by stopping enzymes that degrade it – that’s why you feel happier!
- Ethanol also causes blood vessels to dilate, resulting in flushing of the skin and a sensation of warmth as blood moves into capillaries.
- If you’ve had a large meal, alcohol will stay longer in your stomach, that’s why you can’t get drunk as easily. But eventually, it will enter the bloodstream and do its tricks:)
Your body sees alcohol as an intruder and tries to get rid of it using an enzyme to break it down, “alcohol dehydrogenase“. On contact, the enzyme snatches a hydrogen atom off the ethanol molecules in your drink, rendering it into non-intoxicating acetaldehyde, which is associated with hangovers. Alcohol dehydrogenase is less efficient in Asian populations. Lungs and liver also remove 10% of the alcohol in your body by urine and breath.
Click on this link for a schematic representation of how alcohol works in your brain.
Chemical
- Fuel: The largest single use of ethanol is as a motor fuel, fuel additive, and as a rocket fuel. Over 20% of cars in Brazil are able to run on 100% ethanol fuel.
- Raw material: Ethanol is an important industrial ingredient and has widespread use as a base chemical for other organic compounds, such as ethyl halides, ethyl esters, diethyl ether, acetic acid.
- Solvent: Ethanol is miscible with water and is a good general purpose solvent. It is found in paints, tinctures, markers, and personal care products such as perfumes and deodorants.
- Preservative: for biological specimens.
Medicine
- Antiseptic: Ethanol is used in antiseptic and some antibacterial soaps and wipes. Ethanol kills organisms by denaturing their proteins and dissolving their lipids and is effective against most bacteria and fungi, and many viruses, but is ineffective against bacterial spores.
- Solvent: As a good solvent, you will find ethanol frequently used in many medicine, such as cough syrups.
- Drug: For therapeutic neurolysis: It is injected proximate to nerve tissues and into spinal subarachnoid spaces to produce degeneration of nerve function (neurolysis) for control of chronic pain.
Poison
A couple of days ago I heard in the news that Amy Winehouse‘s death this summer was the result of alcohol poisoning….
A pathologist told a coroner’s court in north London that alcohol toxicity was the cause of the 27-year-old’s death, with her blood-alcohol levels measured at more than five times the legal limit for driving.
The report said that Amy’s blood alcohol content was 416 mg per decilitre at the time of her death. That’s 0.416% alcohol in the blood and about 9 drinks for a woman 45 kg like Amy. When large amounts of alcohol are consumed in a short period of time, alcohol poisoning can occur. Above a blood alcohol concentration of 0.5%, alcohol depresses nerves that control involuntary actions such as breathing and cardiovascular regulation. Moreover, it can cause hypothermia (low body temperature), hypoglycemia (too little blood sugar) and sever dehydration. These effects can lead to cause seizures, permanent brain damage, and ultimately death. That’s why you should never underestimate the effects of alcohol. And think twice before sipping more than three drinks in a row! Better to be safe than sorry.
Celebrating Marie Curie
7 November 2011 is a special day for Chemistry. It marks the 144th anniversary of Marie Curie’s birthday; 2011 has been designated as the International Year of Chemistry (IYC) by IUPAC and UNESC.
And there’s even more: The IYC 2011 coincides with the 100th anniversary of Marie Curie’s Nobel Prize in Chemistry. France and Poland declared 2011 to be the Year of Marie Curie. To recognise her achievements, Marie Curie has been chosen as the symbol for this year’s 2011 IYC – to celebrate it Google has dedicated today its search page to Marie Curie.
Marie Curie is shown by Google at her work bench – indeed she is known for her vigorous passion for science, her hard work that led her to claim two Nobel prizes, and her enormous contributions to Chemistry and the fight against cancer. Her legacy is lived on in several academic institutions and charities such as the Marie Curie Cancer Care. Below are important facts about the “most inspirational woman in science”:
- Marie Sklodowska Curie was born on 7 November 1867 in Warsaw.
- She was awarded the 1903 Nobel prize in Physics, together with husband Pierre Curie and Henri Becquerel for her “researches on the radiation phenomena”.
- On 8 Nov. 1911, she was awarded the 1911 Nobel prize in Chemistry for “the discovery of radium and polonium, the isolation of radium and the study of radium’s nature and compounds”.
- In 1906 she became the first female Sorbonne Professor.
- She is the first of only two people ever to win the Nobel prize in multiple fields. She was also the first woman ever to win the Nobel Prize.
- Curie promoted the use of the radioactivity for therapeutic purposes.
- In 1914 she helped develop small, mobile X-ray units and joined the war front with her 17-year old daughter Irene to help wounded soldierslocate fractures, bullets, and shrapnel.
- Her daughter Irene won the 1935 chemistry Nobel for her work on artificial radioactivity.
- She founded the Curie Institutes in France and Poland, co-founded the Warsaw Radium Institute, and headed the Pasteur Institute.
Curie helped forever change how the world perceived women in science and set a shining example for the future generations of scientists in that rigorous and determined investigation can lead to remarkable discoveries.
7 November is also a very special day for me as it is also my own birthday. I am unbelievably honored to share a birthday with a female chemist of this caliber. I am continuously inspired by her lifelong dedication and contributions to science. As she very well put it:
Nothing in life is to be feared, it is only to be understood. Now is the time to understand more, so that we may fear less.
–Marie Curie
The biography of Marie Curie was written by her daughter, Eve Curie. From Amazon.com:
From Amazon.com:
This biography chronicles Curie’s legendary achievements in science, including her pioneering efforts in the study of radioactivity and her two Nobel Prizes in Physics and Chemistry. It also spotlights her remarkable life, from her childhood in Poland, to her storybook Parisian marriage to fellow scientist Pierre Curie, to her tragic death from the very radium that brought her fame.
Below are some of the many events with which the IYC2011 has honored Marie Curie:
- A Marie Curie inspired poster exhibition was held at the Research Centre for Materials Science, Nagoya University (July 21st -Aug 31st 2011) in collaboration with The Curie Museum and Curie Institute.
- To celebrate her achievements, a re-enactment play inspired by the life of Marie Curie was performed at the IUPAC World Chemistry Congress in San Juan Puerto Rico by professional actress, Susan Frontczak, which was followed by the award ceremony for the 23 Distinguished Women in Chemistry/Chemical Engineering (August 2nd 2011)
- The Geffen Playhouse in Los Angeles, CA is producing the world premiere of Radiance: The Passion of Marie Curie (Nov.1 through Dec. 11, 2011).
- Marie Curie on Stamps Exhibition @ Postalia, Québec city
- 25 November 2011- MSC-100 closing celebration to be held at the Royal Castle in Warsaw.
You will have an exciting business adventure
Last Monday, TedxAcademy in Athens, Greece, organized an event that I will forever remember. Inspired thinkers, entrepreneurs, and innovators were invited to speak about making a difference in a country that is undergoing historical change. There were many excellent speakers, who talked about this time of rapid change, which amidst its high risk, it also offers the incredible opportunity to transform. I thought that two of them just stood out.
Costas Evripides is a founding member and Genesis Pharma’s Chief Executive Officer. In 2006, Costas was voted as the “Dynamic Entrepreneur of the Year 2006”, in the international competition “Entrepreneur Of The Year”. At the TedxAcademy, he advocated about starting a business in Greece. “You will have an exciting business adventure” he told us, if you follow these key ingredients of a successful startup:
- Passion
- Experience
- Create your our own networking circle
- Differentiation
- Specialization
- Stretched targets
- Flexibility
- Reliability
- Alliance building
- Choosing a strong team
And here’s the things to avoid:
- Be fast but don’t rush
- Appreciate the right timing
- Never give up
- Never rest up
And then, there was Stathis Kalyvas, the Arnold Wolfers Professor of Political Science and Director of the Program on Order, Conflict, and Violence at Yale University. Stathis talked about three key aspects to overcome the crisis:
- Incentive
- Vision for the future
- New social framework
I couldn’t agree more. Inspired by my research, on the drug design sector (ie making novel drugs for human diseases), and these two excellent talks, I believe that there are plenty of opportunities to innovate and move forward utilizing the power of the flourishing Greek Pharma Industry. What Greece needs right now is transformation and growth, which could undoubtedly rise from the pharma industry’s active involvement in Greek pharmaceutical and biotech R&D, exploitation of the underutilized capabilities of Greek researchers ultimately leading to the creation of new, Greek drugs. Our country has extremely talented scientists, who could provide access to a multi-billion euro market: health. Greek Pharma-Academia partnerships could be key for emerging stronger out of the crisis.
TedxAcademy is a nonprofit organization inspired by “TED“, an organization devoted to “Ideas Worth Spreading”. Thank you TedxAcademy for the wonderful opportunity to be part of this event!
“Five” – short films about breast cancer’s impact air tonight
Jen Aniston, Demi Moore, Alicia Keys, Penelope Spheeris & Patty Jenkins direct short films abt breast cancer’s impact.
“Five” airs tonight at mylifetimetv.
The groundbreaking original movie “Five” is an anthology of five short films exploring the impact of breast cancer on people’s lives. “Five” highlights the shared experience each short film’s title character endures from the moment of diagnosis, through an interconnected story arc that uses humor and drama to focus on the effect breast cancer and its different stages of diagnosis have on relationships and the way women perceive themselves while searching for strength, comfort, medical breakthroughs and, ultimately, a cure.
What is breast cancer?
Your cells have a normal cycle of birth, life, and death. Normally, new cells are generated only when they are needed. When the cells stop dying and replicate uncontrollably, they create a mass of tissue, which is called a tumor. If the cells that are growing out of control are normal cells, the tumor is called benign (not cancerous). If however, the cells that are growing out of control are abnormal and don’t function like the body’s normal cells, the tumor is called malignant (cancerous).
Therefore, cancer is not a disease caused by an external organism, such as a virus or bacteria, but they are your own cells growing uncontrollably and obstructing the function of normal tissues and organs.
Cancers are named after the part of the body from which they originate, so breast cancer originates from breast cancer cells. Like other cancers, breast cancer cells can invade surrounding tissues and also travel to other parts of the body and form new tumors, a process called metastasis.
Facts about breast cancer
Here is some data taken from here and here:
- Breast cancer is the most common cancer among American women, after skin cancer.
- Breast cancer is the second-leading cause of cancer death in women after lung cancer — and is the leading cause of cancer death among women ages 35 to 54.
- Today, approximately 1 in almost every 8 women (13.4%) will develop breast cancer in her lifetime.
- Only 5% to 10% of breast cancers occur in women with a clearly defined genetic predisposition for the disease. The majority of breast cancer cases are “sporadic,” meaning there is no direct family history of the disease.
- The risk for developing breast cancer increases as a woman ages.
- $13.9 billion is spent every year in breast cancer treatments in the US alone.
- And the good news: There are more than 2.5 million breast cancer survivors living in the US.
- Women can lower their risk of breast cancer by lowering alcohol intake, exercising regularly, and maintaining a healthy wait. Getting regular screens can result in early detection, which increases the chance of survival.
Many thanks to the makers of this film for showing that breast cancer awareness is so much more than wearing a pink ribbon!!!
Be an advocate and get screened. Do self exams. Donate to cancer research. Learn what your own risks are and how best to prevent or minimize your risks. We can all make a real difference.
How is coffee decaffeinated?
Apparently it’s National Coffee Day today, read below to learn in less than a page how coffee gets decaffeinated – not an easy task!
Coffee contains over 400 chemicals important to the taste and aroma of the final drink: it is therefore challenging to remove only caffeine while leaving the other chemicals at their original concentrations.
 To get rid of the caffeine, unroasted (green) beans are at first steamed. The beans are then rinsed with a “solvent” chemical that extracts the caffeine while leaving the other essential chemicals in the coffee beans. The process is repeated anywhere from 8 to 12 times until caffeine is removed in 97%-99.9%. So what is this magic “solvent” chemical. which is able to selectively remove caffeine while preserving the coffee aroma?
To get rid of the caffeine, unroasted (green) beans are at first steamed. The beans are then rinsed with a “solvent” chemical that extracts the caffeine while leaving the other essential chemicals in the coffee beans. The process is repeated anywhere from 8 to 12 times until caffeine is removed in 97%-99.9%. So what is this magic “solvent” chemical. which is able to selectively remove caffeine while preserving the coffee aroma?
This solvent is nothing else but carbon dioxide (CO2), which is also found in our very own atmosphere. However, as you might have guessed, CO2 is not used for decaffeination as a gas from the atmosphere. For the decaffeination process, the coffee beans are soaked in a special form of CO2 called “supercritical” CO2. OK, so now you naturally have to ask what the heck is this “supercritical” CO2??
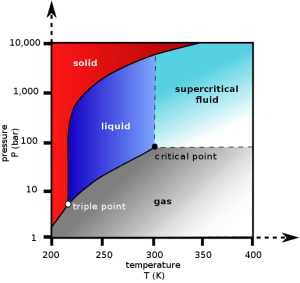
Every substance (water, hydrogen, oxygen, etc) has a so-called “critical point”. The critical point is the point above which, the distinction between liquid and gas phases of that substance stops being meaningful. That is you cannot tell any more whether this substance is gas or liquid. This new “phase”, being neither gas or liquid is called “supercritical”. A “supercritical fluid” can effuse through solids like a gas, and dissolve materials like a liquid.
So how does supercritical CO2 look like?
To achieve the supercritical state for most substances requires extreme temperatures and pressures. Carbon dioxide, however, has a fairly accessible critical point at ~ 31.1°C and 73 atm (our atmosphere has 1 atm). And it seems that supercritical CO2 has unique properties such that it can selectively extract caffeine from coffee beans, producing decaf coffee.
Carbon dioxide pressure-temperature phase diagram. The critical point is found at T=31.1°C and 73 atm. Above the critical point, one cannot really tell whether CO2 is liquid or gas. This new phase is called “supercritical”.