Author Archives: zoecournia
Small/Medium Enterprises (SMEs) in Horizon 2020
“SMEs- The new tools/opportunities for access to finance introduced by Horizon 2020” by Jean-David Malo, Head of Unit SMEs, Financial Instruments and State Aid, Directorate General for Research and Innovation, EC.
“Access to risk finance”
Part of the horizon 2020 budget, that is 3,69% (2.7 billion euros) will be provided though grant funding but in the form of risk sharing for loans and guarantees and by providing risk finance (equity).
Goal: stimulate more investment in research and innovation To attract additional finance and multiply horizon 2020 budget resources for SMEs
Why?
Because of enduring difficulties in SMEs in accessing startup/Grant finance and because of the need to build an integrated European VC market
They cover a broad range of interventions such as :
- Guarantees to mutual guarantee societies and banks lending to companies incl SMEs
- Equity participation for early and growth stage investments
This funding will be typically delivered via financial intermediaries (banks funds etc) but also directly Operated by the EIB group but may also be operated by other financial institutions
Debt finance can be achieved through the following schemes:
- Loans service for R&I (called “RSFFII”) Loans and guarantees for investments in research and innovation; targeted at midcaps and larger companies, research institutes and Stand alone projects Loan amounts over 7,5 million
- Sme and small midcaps R&I loans service Loan guarantee facility between 25,000 to 7.5 million
- SME initiative : Joint guarantee instrument an securitization for loans to innovative SMEs and small midcaps
Equity finance (next year):
-Early stage finance for innovative enterprises
-Pilot facility for tech transfer , creation of spin offs
These schemes will be facilitated by the Horizon 2020 along with the “COSME” program.
More info on the initiatives can be also found here.
For the full presentation of Horizon 2020’s Financial Instruments, check this link.
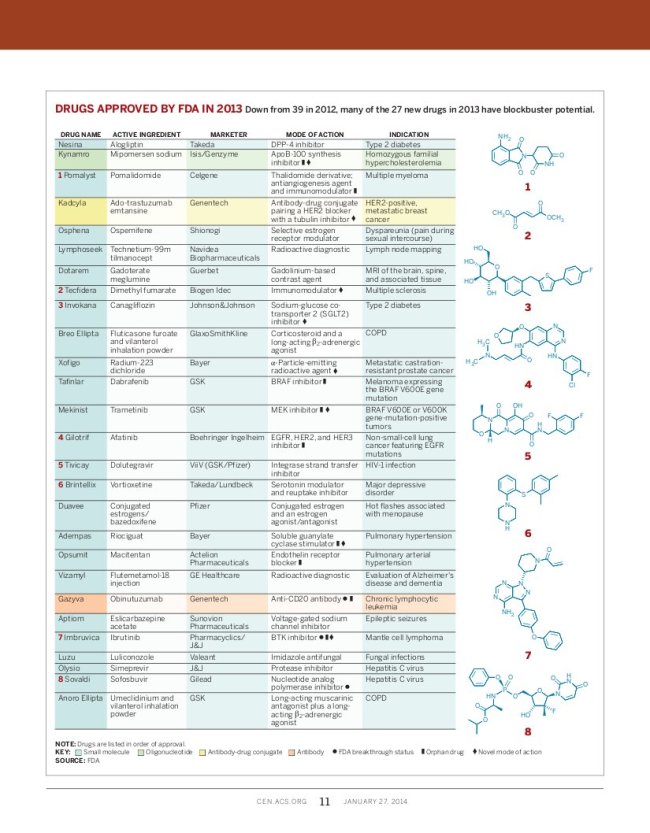
FDA Approves 27 New Drugs in 2013
The new drug approvals for 2013 were mostly cancer drugs and drugs for orphan diseases.
Other therapeutic areas included metabolic and endocrinological therapies (two type 2 diabetes drugs and one dyslipidaemia drug), antivirals (two hepatitis C virus (HCV) drugs and an HIV drug) and medical imaging products (three approvals).
Highlights
Several of these new drugs are scientifically novel.
Gilead’s sofosbuvir is first-in-class NS5B polymerase inhibitor and the first oral HCV drug (Nature Rev. Drug Discov. 12, 409–411; 2013).
Pharmacyclics and Janssen’s ibrutinib is a first-in-class Bruton’s tyrosine kinase (BTK) inhibitor, which entered the clinic in 2009, and is now approved for an aggressive form of B cell non-Hodgkin’s lymphoma, but it is now being also tested for other types of cancers such as chronic lymphocytic leukaemia (CLL). Ibrutinib is the second irreversible kinase inhibitor to be approved after afatinib, setting new hope for the development of irreversible agents that have been thought difficult to reach the market in earlier years (Nature Rev. Drug Discov. 12, 649–651; 2013).
Biogen Idec’s dimethyl fumarate (formerly BG-12) is an extremely small molecule (MW only 144 Da) that had been approved as an anti-psoriasis drug in Germany as well as for preventing the growth of mold in furniture! Now Biogen Idec gets its approval for dimethyl fumarate as the third oral drug for multiple sclerosis. Drug mechanism remains unclear.
GSK’s trametinib is a first-in-class MAPK/ERK kinase (MEK) inhibitor that overcame bioavailability, pharmacokinetics and toxicity problems of previous drug candidates (Nature Rev. Drug Discov. 11, 819–820; 2012). Trametinib is approved for cancers bearing specific BRAF mutations (such as metastatic melanoma). GSK picked up five approvals from the FDA last year (counting the approval granted to ViiV), more than any other drug developer in 2013.
Janssen’s canagliflozin, is the first-FDA approved sodium-dependent glucose cotransporter 2 (SGLT2) inhibitor, against type 2 diabetes space.
Genzyme and Isis’s mipomersen acts on a novel target, binding apolipoprotein B mRNA, reducing protein synthesis and thereby lowering low-density lipoprotein (LDL) levels. Because of its side effects it may be used only as an orphan disease drug agains homozygous familial hypercholesterolaemia and not as a general cholesterol-lowering agent. Regulators in the European Union (EU) have rejected mipomersen because of its side-effects.
Sources:
Why naked mole rats never get cancer
 Arguably this is not the most adorable creature you have ever seen. But, the naked mole rat (Heterocephalus glaber) displays exceptional longevity, with a maximum lifespan exceeding 30 years. In human years (relative to body size as naked mole rats) that is 600 years!
Arguably this is not the most adorable creature you have ever seen. But, the naked mole rat (Heterocephalus glaber) displays exceptional longevity, with a maximum lifespan exceeding 30 years. In human years (relative to body size as naked mole rats) that is 600 years!
In addition to their longevity, naked mole rats, these subterranean African mammals, show an unusual resistance to cancer. Multi-year observations of large naked mole-rat colonies did not detect a single incidence of cancer! The authors found that the extracellular matrix in naked mole rats is rich in a substance that stops cancers growing. The magic ingredient is a polysaccharide called hyaluronan, which acts as a lubricant in the body. This particular hyaluronan is an extremely high-molecular-mass hyaluronan (HA), which is over five times larger than human or mouse HA. 
By manipulating the pathways that lead to the build-up of high-molecular-mass hyaluronan in cells, the authors showed that if we prevent the naked mole rats from making high-molecular-mass hyaluronan then tumours can be grown.
Biotechs and SMEs will be allocated 20% of the overall budget in the Horizon 2020 scheme
While the new 7-year funding scheme of EU, “Horizon 2020” will continue to fund excellent basic research (17% of the total budget), an effort is being made to attract small and medium enterprises (SMEs) to participate in research consortia. After a two-and-a-half-year negotiation, the European Council and the European Parliament reached an agreement (pending final sign-off) that SMEs will claim a 20% of the total budget in an act that aims to boost European economy.
Open access publication of results of all research funded under Horizon 2020 will be mandatory and awards will be made within 8 months as opposed to 12 months, which is the current scheme.
Industry – academia partnerships: way to go for anticancer drug development?
Industry – academia partnerships are increasingly growing in an effort to make the drug discovery process more efficient. In a Cancer Research UK perspective, several industrial collaborations are outlined with an eye on the distinct but com-plementary expertise brought about in anti-cancer drug development.
For more industry – academia initiatives check out
GlaxoSmithKline’s Discovery Fast Track competition for academic researchers
Eli Lilly’s Open Innovation Platform
Marie Curie’s Industry-Academia Partnerships and Pathways (IAPP)
Read another article on industry – academia partnerships from Science Magazine and an older article from this blog on the topic here.
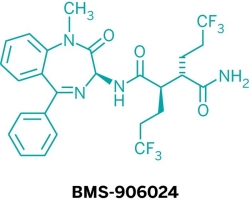
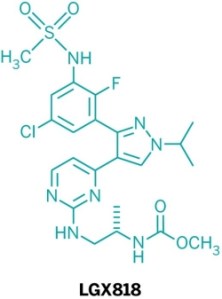
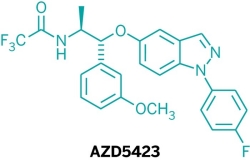
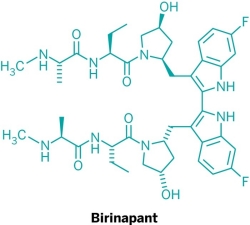
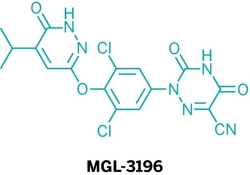
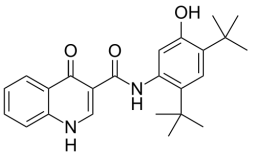
Five new candidate drugs are revealed in the ACS New Orleans meeting
The ACS National Spring Meeting took place 7-11 April in New Orleans.
As is traditional for the Medicinal Chemistry Division, structures of candidate drugs get revealed in the “First-Time Disclosures” session.
This year’s entries are:
More about how where these drugs discovered:http://cen.acs.org/articles/91/i16/Five-New-Drug-Candidates-Structures.html
Kalydeco: The most important new drug of 2012
Kalydeco, a drug for cystic fibrosis, is the most important new drug of 2012 according to Forbes magazine and was developed by Vertex pharmaceuticals with seed funding from the Cystic Fibrosis Foundation.
Cystic Fibrosis is a genetic disorder that results in scarring (fibrosis) and cyst formation within the pancreas, lungs, liver, and intestines.
Kalydeco, given alone, will only help a few thousand patients the world over. Like other drugs for very rare diseases, its price is very high: $294,000 per patient per year.
Though its chemical structure could be routinely made by a synthetic chemist, it is covered by a patent so it is illegal to make in a lab.
The efforts to cure cystic fibrosis were spearheaded by a discovery from Francis Collins, later famous for heading the Human Genome Project and then the National Institutes of Health, who discovered the gene that, when mutated, causes cystic fibrosis 23 years ago. Kalydeco is the first drug to directly affect the defects caused by these mutations, leading to improvements in patients’ lung function.
366 Days: The Year in Science
Read below the Science Review of 2012 by Nature Magazine, with Greece making it to the top 22 “leading science nations” with 1% of the ‘most cited papers’!
Also, read on Science Magazine’s Breakthrough of the Year 2012 (Higgs Boson) and the runners-up: Genome Engineering, Curiosity Landing, Bionics, Eggs from Stem Cells, Encode, X-ray laser advances and more!
http://www.sciencemag.org/site/special/btoy2012/
2012 in review
The WordPress.com stats helper monkeys prepared a 2012 annual report for Life is Chemistry.
Here’s an excerpt:
600 people reached the top of Mt. Everest in 2012. This blog got about 4,900 views in 2012. If every person who reached the top of Mt. Everest viewed this blog, it would have taken 8 years to get that many views.
The Chemistry of Euro banknotes
Europium(III) oxide, alongside other chemicals, is phosphorescent and is used in the anti-counterfeiting details in Euro banknotes.
 Europium (symbol Eu) is one of the rare earth elements and belongs to the class of “lanthanides”.
Europium (symbol Eu) is one of the rare earth elements and belongs to the class of “lanthanides”.
Most of the trivalent rare earth elements are luminescent. This means that they can be excited by shining a light of a particular wavelength at them. When the ions relax again, they emit light – of a different wavelength. That is luminescence.
Euro notes luminesce in the red, green and blue (excited by 254 nm). The red light is clearly linked to europium and most likely to a Eu3+-β-diketone complex.